Table of Contents
Introduction
Lung cancer is a leading cause of cancer-related deaths worldwide. Of its types, non-small cell lung cancer (NSCLC) accounts for nearly 85% of all lung cancer cases. Over the past decade, understanding the molecular pathways involved in cancer progression has opened new doors for targeted therapies.
One of the most exciting advances in this field is sunvocertinib, sold under the brand name Zegfrovi. This drug has brought new hope to patients with locally advanced or metastatic NSCLC with epidermal growth factor receptor (EGFR) mutations.
Sunvocertinib is an oral, potent, irreversible, and mutation-selective EGFR tyrosine kinase inhibitor (TKI) designed to overcome resistance mechanisms that have limited the effectiveness of previous EGFR-targeted drugs.
What Is Sunvozertinib?
Sunvocertinib (Zegfrovi) belongs to a class of targeted anti-cancer therapies known as EGFR tyrosine kinase inhibitors (TKIs). These drugs are specifically designed to target mutated forms of the EGFR gene, which drive the uncontrolled growth of cancer cells in NSCLC.
It is unique in its ability to selectively inhibit mutated EGFR forms with EGFR exon 20 insertion mutations, a notoriously difficult target that resists many traditional EGFR inhibitors.
🔹 Key Highlights:
- Brand Name: Zegfrovy
- Generic Name: Sunvozertinib
- Drug Class: EGFR Tyrosine Kinase Inhibitor (TKI)
- Dosage Form: Oral tablet
- Indication: Locally advanced or metastatic NSCLC with EGFR exon 20 insertion mutations
- Mechanism: Irreversible and mutant-selective inhibition of EGFR signaling
Mechanism of Action:
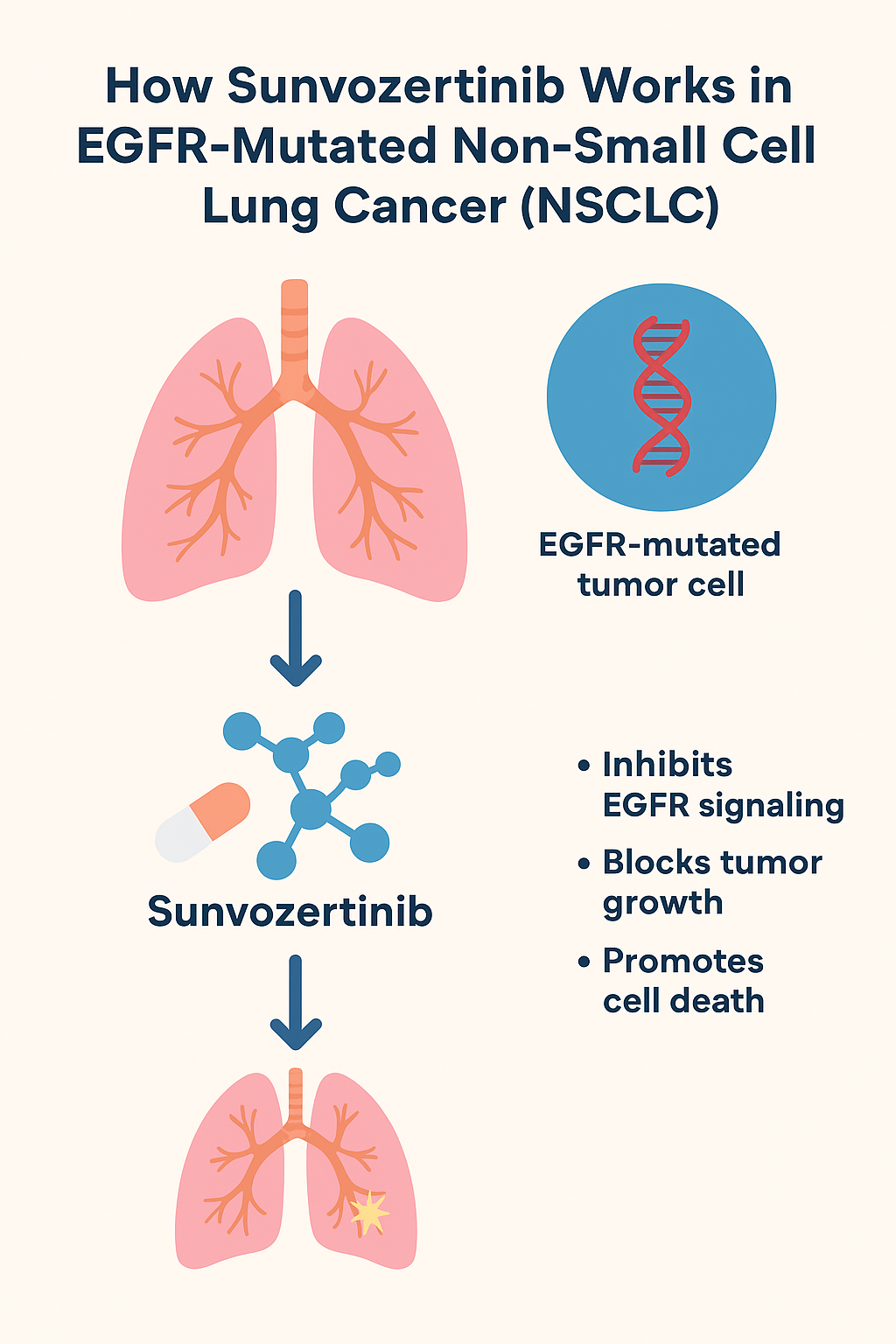
To understand how sunvocertinib works, let’s look at its molecular mechanism.
The epidermal growth factor receptor (EGFR) is a protein found on the surface of many cells. When activated by its ligands, it initiates a cascade of signals that regulate cell growth and division. In many cases of NSCLC, mutations in the EGFR gene cause the receptor to become permanently activated, leading to uncontrolled cell proliferation – a hallmark of cancer.
Sunvocertinib binds irreversibly to the mutated EGFR enzyme, blocking its kinase activity. In doing so, it effectively stops the growth of cancer cells, promotes apoptosis (programmed cell death), and helps shrink tumors.
⚡ Step-by-Step Mechanism:
- Binding to Mutant EGFR:
Sunvozertinib specifically targets the altered EGFR receptor sites found in cancer cells. - Irreversible Inhibition:
It covalently binds to the kinase domain, permanently inactivating EGFR signaling. - Signal Suppression:
This stops downstream pathways such as RAS-RAF-MEK-ERK and PI3K-AKT, which are responsible for cell survival and proliferation. - Tumor Growth Control:
Without these signals, cancer cells undergo apoptosis, leading to tumor shrinkage and delayed disease progression.
Clinical Significance in NSCLC
Traditional chemotherapy targets both healthy and cancerous cells, often leading to severe side effects. In contrast, Sunvozertinib offers a precision-based approach, selectively attacking tumor cells that harbor EGFR exon 20 insertion mutations — one of the most aggressive and treatment-resistant forms of NSCLC.
✅ Advantages of Sunvozertinib in NSCLC:
- Effective in EGFR exon 20 insertion mutations
- Improved progression-free survival (PFS)
- High response rates in both treatment-naïve and previously treated patients
- Fewer off-target effects compared to earlier TKIs
- Oral administration for better patient compliance
This selectivity and oral dosing make Sunvozertinib a preferred therapy in eligible NSCLC cases, enhancing both survival and quality of life.
Pharmacokinetics of Sunvozertinib
Understanding how Sunvozertinib behaves inside the body provides insight into its effectiveness and safety.
| Parameter | Description |
| Absorption | Rapidly absorbed after oral administration. |
| Distribution | Widely distributed into tumor tissues and plasma. |
| Metabolism | Metabolized primarily in the liver through CYP3A4 enzymes. |
| Elimination Half-Life | Approximately 30–40 hours. |
| Excretion | Mainly through feces, minimal renal clearance. |
This long half-life allows once-daily dosing, making it convenient for chronic cancer management.
Dosage and Administration
The recommended Sunvozertinib (Zegfrovy) dosage is taken orally once daily, with or without food. The dosage may vary depending on patient tolerance, concurrent medications, and specific clinical conditions.
Patients should swallow tablets whole and not crush or chew them. Regular monitoring of liver function, complete blood count, and ECG is advised during treatment.
Note: Dosage adjustments may be necessary in case of severe adverse reactions or hepatic impairment.
Sunvozertinib Side Effects
Like most cancer therapies, Sunvozertinib can cause side effects, although its selective mechanism reduces the risk of widespread toxicity.
Common Side Effects:
- Diarrhea
- Rash or acneiform eruption
- Fatigue
- Decreased appetite
- Nausea
- Dry skin
Serious Adverse Effects:
- Interstitial lung disease (ILD) / pneumonitis
- QT interval prolongation (affecting heart rhythm)
- Hepatotoxicity (liver damage)
- Severe diarrhea leading to dehydration
Patients experiencing severe respiratory or cardiac symptoms should seek medical attention immediately.
Regular follow-up with oncologists helps adjust therapy and minimize complications.
Drug Interactions
Sunvozertinib interacts with medications that influence CYP3A4 activity.
Avoid:
- Strong CYP3A4 inducers (e.g., rifampin, phenytoin, carbamazepine)
- Strong CYP3A4 inhibitors (e.g., ketoconazole, clarithromycin)
- Grapefruit juice, as it can alter plasma concentrations
Co-administration with other QT-prolonging drugs should also be approached cautiously.
Clinical Trials and Research Findings
Clinical trials have demonstrated Sunvozertinib’s remarkable efficacy in patients with advanced NSCLC carrying EGFR exon 20 insertion mutations.
In pivotal studies, Sunvozertinib achieved:
- Overall response rate (ORR): Over 50%
- Median progression-free survival (PFS): Around 8–10 months
- Durable responses even after prior TKI or chemotherapy failure
Additionally, patients reported improvements in quality of life and symptom management.
Its mutant-selective design ensures reduced off-target toxicity compared to earlier-generation EGFR inhibitors.
These outcomes have positioned Sunvozertinib as a next-generation therapy in precision oncology.
Global Approval and Regulatory Status
Sunvozertinib (Zegfrovy) is approved in several regions for:
- Treatment of adult patients with locally advanced or metastatic NSCLC
- Presence of EGFR exon 20 insertion mutations
- Progression on or after platinum-based chemotherapy
The U.S. FDA, European Medicines Agency (EMA), and other global health authorities have recognized its clinical benefits in targeted cancer therapy.
This approval marks a significant advancement in the treatment landscape for NSCLC, offering a life-extending option for patients previously left with limited choices.
Benefits of Sunvozertinib Over Other EGFR TKIs
| Parameter | First/Second Generation TKIs | Sunvozertinib (Zegfrovy) |
| Mutation Targeting | Common EGFR mutations | EGFR exon 20 insertions |
| Binding Type | Reversible | Irreversible |
| Selectivity | Non-selective | Mutant-selective |
| Resistance Handling | Limited | Overcomes resistance |
| CNS Penetration | Moderate | Improved |
| Adverse Effects | Broader | More controlled |
This table highlights how Sunvozertinib provides superior selectivity, durability, and safety, making it a crucial therapy for mutation-specific NSCLC.
Patient Counseling and Education
Patients prescribed Sunvozertinib should be educated about:
- The importance of daily adherence
- Reporting side effects early
- Avoiding grapefruit and certain medications
- Regular blood and ECG monitoring
- Staying hydrated and maintaining nutrition during therapy
Educating patients enhances compliance, reduces adverse effects, and ensures optimal outcomes.
Future Prospects
Ongoing clinical trials are exploring Sunvozertinib in combination with immunotherapy, chemotherapy, and other TKIs to further enhance its therapeutic impact.
Researchers are also investigating its potential for brain metastases, as Sunvozertinib demonstrates strong CNS penetration, a critical advantage in NSCLC patients with brain involvement.
This continued innovation may expand its indications to earlier lines of therapy or adjuvant settings, improving survival across broader patient populations.
Conclusion
Sunvocertinib (Zegfrovi) represents an ideal change in the treatment of EGFR-mutated NSCLC, particularly those with exon 20 insertion mutations, which have historically been resistant to standard TKIs.
Its mutant-selective, irreversible mechanism of action provides a powerful yet precise therapeutic option, improving both clinical outcomes and quality of life for patients.
As research evolves, sunvocertinib stands as a beacon of innovation in precision oncology – paving the way for smarter, safer, and more effective cancer care.