Table of Contents
Introduction: A Milestone in Allergy Treatment
On September 30, 2025, the US Food and Drug Administration (FDA) approved remibrutinib, marketed under the brand name Rapsido, for adult patients with chronic spontaneous urticaria (CSU) who are not responding to treatment with H1 antihistamines.
This approval is a significant milestone, as remibrutinib becomes the first oral Bruton’s tyrosine kinase (BTK) inhibitor approved for CSU. It offers a new, targeted therapy for the millions of people who suffer from persistent hives and itching without a clear cause.
In this article, we will explore the remibrutinib FDA approval, its mechanism of action, dosage, clinical trial results, side effects, and patient outcomes, allowing for an in-depth review of this breakthrough treatment.
What Is Chronic Spontaneous Urticaria (CSU)?
Chronic spontaneous urticaria, also known as chronic idiopathic urticaria, is a skin condition characterized by itchy, red, swollen hives that persist for six weeks or longer without any identifiable trigger.
Symptoms can come and go unpredictably, and although H1 antihistamines are the standard first-line therapy, nearly half of patients do not experience complete relief of symptoms.
CSU can severely impact a person’s sleep quality, emotional well-being, and daily functioning. Prior to the Remibrutinib FDA Approval, treatment options were limited to antihistamines and injectable biologics such as omalizumab, which required regular injections and were expensive for long-term use.
Remibrutinib FDA Approval: A Game-Changer
The Remibrutinib FDA Approval (Rhapsido) represents a paradigm shift in CSU management. Unlike traditional therapies, Remibrutinib targets the root immunological mechanism of the disease rather than just treating symptoms.
- Approval Date: September 30, 2025
- Brand Name: Rhapsido
- Manufacturer: Novartis Pharmaceuticals
- Indication: For adult patients with chronic spontaneous urticaria who remain symptomatic despite H1 antihistamines
- Drug Class: Oral Bruton’s Tyrosine Kinase (BTK) Inhibitor
This approval was based on robust phase 3 clinical trial data, showing significant reductions in itch severity and hive count compared to placebo.
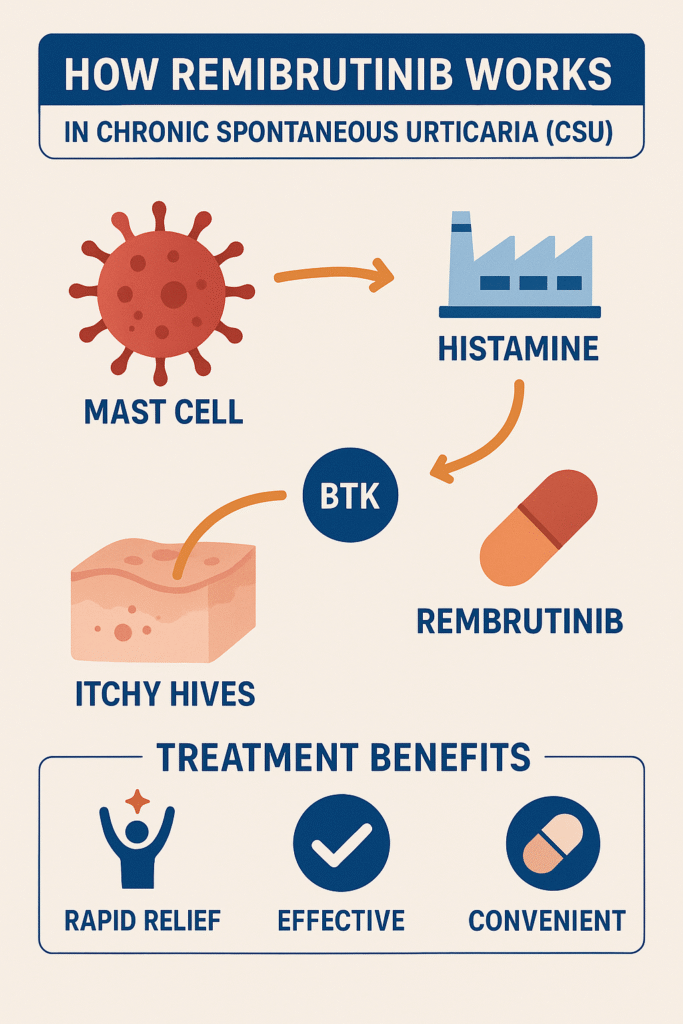
Mechanism of Action: How Remibrutinib Works
Remibrutinib belongs to a class of drugs known as Bruton’s Tyrosine Kinase inhibitors (BTKi).
BTK plays a critical role in the activation of mast cells and basophils, which are immune cells responsible for releasing histamine and other inflammatory mediators that cause itching, redness, and swelling.
Key Mechanism Steps:
- BTK inhibition: Remibrutinib selectively binds to and inhibits BTK.
- Reduced immune cell activation: This prevents the activation of mast cells and basophils.
- Decreased histamine release: As a result, inflammatory mediators that trigger hives and itching are reduced.
- Symptom control: Patients experience fewer hives, less itching, and improved quality of life.
By interrupting the immune signaling pathway upstream, Remibrutinib offers a novel, oral, non-biologic therapy for CSU — a major advancement over injectables.
Clinical Trials Supporting Remibrutinib FDA Approval
The FDA’s decision was backed by data from the REMIX-1 and REMIX-2 phase 3 clinical trials. These global studies evaluated Remibrutinib in patients with inadequately controlled CSU despite H1 antihistamines.
Trial Overview:
- Study Design: Randomized, double-blind, placebo-controlled
- Participants: Over 1,000 adults with moderate to severe CSU
- Duration: 24 weeks
- Primary Endpoint: Reduction in weekly itch severity score (ISS7)
- Secondary Endpoint: Decrease in hive count (HSS7) and improvement in quality-of-life scores
Results:
- Patients treated with Remibrutinib achieved a rapid reduction in itch as early as Week 2.
- By Week 12, many achieved complete or near-complete hive resolution.
- Adverse effects were generally mild to moderate, with most patients tolerating therapy well.
These promising results highlight Remibrutinib’s efficacy, safety, and rapid onset of action, leading to its historic FDA approval.
Dosage and Administration
- Formulation: Oral tablet
- Recommended Dose: 50 mg twice daily (as per clinical trial protocols)
- Route: Oral
- Duration: As directed by physician based on response and tolerance
Unlike injectable biologics, Remibrutinib offers the convenience of oral administration, improving treatment adherence and patient comfort.
Side Effects of Remibrutinib
While Remibrutinib is generally well tolerated, like any medication, it can cause side effects. Most are mild and manageable.
Common Side Effects:
- Headache
- Diarrhea
- Fatigue
- Nausea
- Upper respiratory tract infections
Serious Side Effects (Rare):
- Liver enzyme elevation
- Increased risk of infections (due to immune modulation)
- Hypersensitivity reactions
Patients should undergo regular monitoring during therapy and report any unusual symptoms to their healthcare provider.
Remibrutinib vs. Existing CSU Treatments
Before Remibrutinib FDA approval, most CSU patients relied on:
- H1 antihistamines (first-line treatment)
- Omalizumab (Xolair) – an injectable biologic for refractory CSU
- Cyclosporine – an immunosuppressant used off-label
| Feature | Antihistamines | Omalizumab | Remibrutinib |
| Route | Oral | Injection | Oral |
| Onset of Action | Slow | Moderate | Fast |
| Mechanism | Blocks histamine receptors | Binds IgE antibodies | Inhibits BTK pathway |
| Convenience | High | Moderate | Very High |
| Cost | Low | High | Moderate |
| Effectiveness | Limited in refractory cases | Strong | Strong + Convenient |
Thus, Remibrutinib (Rhapsido) offers an effective, oral, and patient-friendly alternative to injections — a true innovation in CSU therapy.
Patient Benefits: Why Remibrutinib Matters
The Remibrutinib FDA Approval brings hope to thousands living with uncontrolled CSU. Its key benefits include:
✅ First oral BTK inhibitor for CSU
✅ Rapid symptom relief – noticeable improvements within weeks
✅ Convenient oral dosing (no injections)
✅ Well-tolerated with manageable side effects
✅ Improved quality of life and sleep
With this new treatment option, patients can regain control over their symptoms and enjoy greater freedom in daily life.
Future Outlook: Expanding BTK Inhibitor Therapies
The success of the Remibrutinib FDA Approval could pave the way for BTK inhibitors in other allergic and autoimmune diseases.
Research is ongoing in asthma, atopic dermatitis, and food allergy, where BTK inhibition may play a key role in reducing hypersensitivity reactions.
The future of allergy medicine is shifting toward precision immunomodulation, and remibrutinib is leading that transformation.
Conclusion: A New Era for CSU Treatment
The Remibrutinib FDA Approval is a revolutionary step forward in the treatment of chronic spontaneous urticaria. For the first time, patients can effectively manage their condition with an oral, targeted therapy that directly targets the immune system. With its proven efficacy, favorable safety profile, and patient-friendly administration, remibrutinib (Rapcid) stands as a landmark innovation – offering new hope for millions of people struggling with chronic urticaria.

1 thought on “Remibrutinib FDA Approval: A New Hope for Chronic Spontaneous Urticaria (CSU)”