Table of Contents
Pivekimab Sunirine – A Complete 2025 Guide for Pharmacy Students & Healthcare Professionals
Pivekimab Sunirin is one of the most advanced targeted therapies developed for hematologic malignancies, particularly relapsed or refractory acute leukemia. As oncology evolves rapidly, this drug has attracted attention due to its unique mechanism of action, CD123 specificity, and promising clinical activity in difficult-to-treat cases.
This comprehensive guide covers everything you need to understand about Pivekimab Sunirin – mechanism of action, pharmacokinetics, clinical trial data, indications, side effects, dosage, counseling issues, and future prospects.
This article is written in simple, high-quality, student-friendly language that is suitable for B. Pharm, D. Pharm, GPAT aspirants, physicians, pharmacists, and cancer-care educators.
What Is Pivekimab Sunirine? – Introduction to the Drug
Pivekimab Sunirin (also known as IMGN632) is a novel antibody-drug conjugate (ADC) designed to target CD123, a biomarker highly expressed in several blood cancers.
It combines:
• A humanized monoclonal antibody targeting CD123
• A potent DNA-alkylating payload
• A cleavable linker that delivers the cytotoxic agent directly to cancer cells
This design enhances anti-tumor activity while minimizing toxicity to healthy tissue.
Why Pivekimab Sunirin is important
• It is specifically designed for relapsed or refractory leukemia, where treatment options are extremely limited.
• Helps reduce systemic toxicity compared to conventional chemotherapy.
• Has shown significant activity in BPDCN, AML, and other CD123-expressing malignancies.
• Represents a major advance in targeted hematologic therapy.
Focus Keyword Relevance – Why Pivekimab Sunirine Matters in 2025
Pivekimab Sunirin has become one of the most sought-after oncology drug candidates because:
• It addresses an unmet clinical need in highly aggressive cancer types.
• It is part of a growing trend in targeted ADC therapies.
• The need for healthcare professionals to understand CD123-targeted therapies is increasing.
• Current trials in 2024-25 show expanded indications and improved outcomes.
As such, pharmacy and medical students should thoroughly learn this molecule for exams, interviews, and clinical practice.
Chemical Class & Pharmacology of Pivekimab Sunirine
Drug Class
- Antibody–Drug Conjugate (ADC)
- CD123-directed therapy
- Alkylator payload-based cytotoxic agent
Structure Components
Pivekimab Sunirine consists of three essential parts:
1. Targeting Antibody
Humanized IgG monoclonal antibody that selectively binds to CD123.
2. Linker
A cleavable peptide linker that remains stable in systemic circulation but releases the payload inside cancer cells.
3. Payload
An indolinobenzodiazepine pseudodimer (IGN), which causes potent DNA cross-linking and apoptosis.
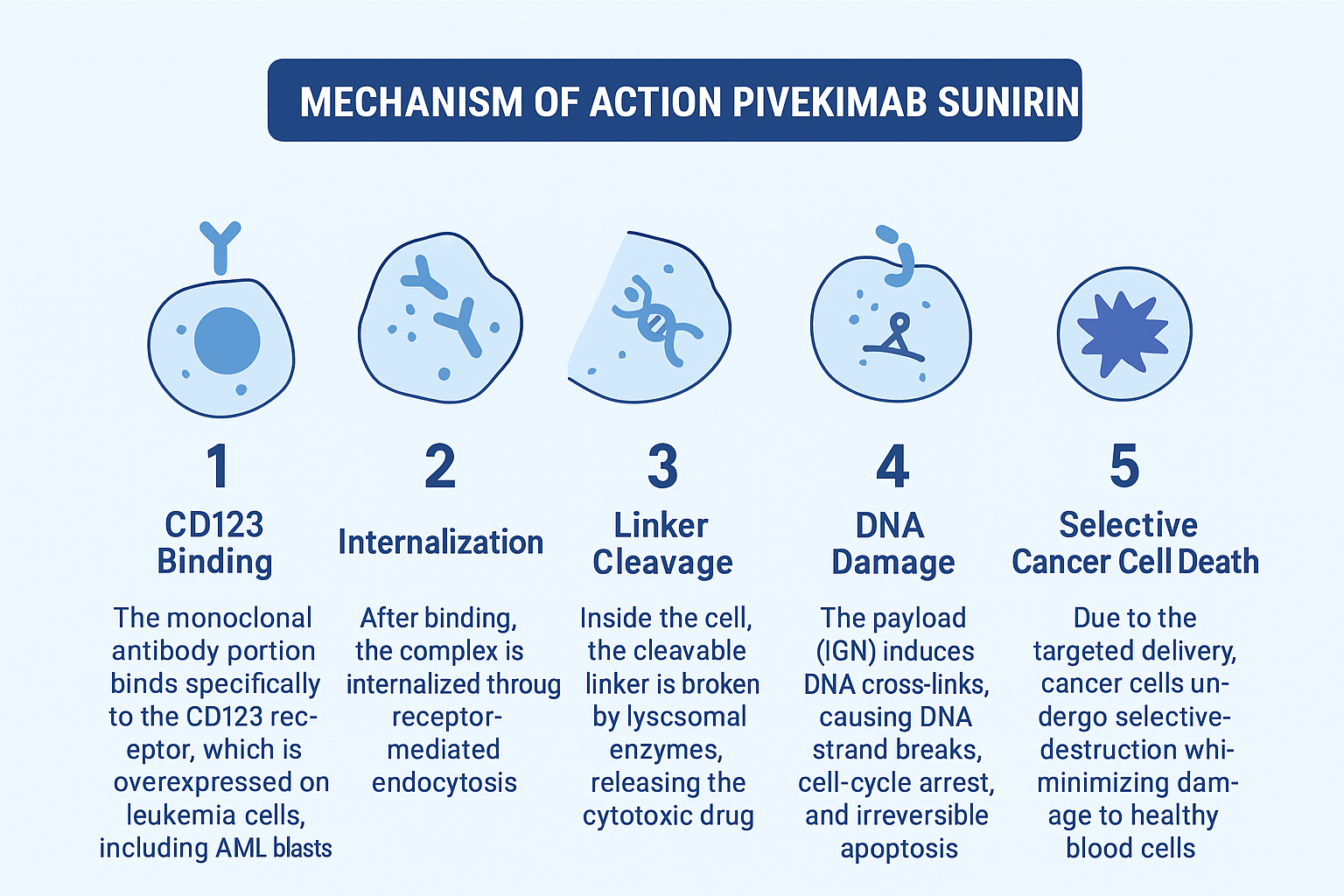
Mechanism of Action of Pivekimab Sunirine (Detailed Step-by-Step)
Step 1 – CD123 Binding
The monoclonal antibody portion binds specifically to the CD123 receptor, which is overexpressed on leukemia cells, including:
- AML blasts
- BPDCN cells
- Leukemic stem cells
Step 2 – Internalization
After binding, the complex is internalized through receptor-mediated endocytosis.
Step 3 – Linker Cleavage
Inside the cell, the cleavable linker is broken by lysosomal enzymes, releasing the cytotoxic drug.
Step 4 – DNA Damage
The payload (IGN) induces DNA cross-links, causing:
- DNA strand breaks
- Cell-cycle arrest
- Irreversible apoptosis
Step 5 – Selective Cancer Cell Death
Due to the targeted delivery, cancer cells undergo selective destruction while minimizing damage to healthy blood cells.
Indications – What Conditions Does Pivekimab Sunirine Treat?
As of 2025, Pivekimab Sunirine is primarily studied or used in:
1. BPDCN (Blastic Plasmacytoid Dendritic Cell Neoplasm)
One of the rarest and most aggressive blood cancers, heavily dependent on CD123 expression.
2. Acute Myeloid Leukemia (AML) – Relapsed/Refractory
Promising response rates seen in several clinical trials.
3. MRD-positive AML
Being explored for minimal residual disease.
4. Combination therapy in newly diagnosed AML
Trials with azacitidine + venetoclax + Pivekimab Sunirine show improved outcomes.
5. High-risk Myelodysplastic Syndromes (MDS)
Investigational use in CD123-positive cases.
Clinical Trial Details – Evidence Supporting
Phase I/II Studies
Clinical trials demonstrate:
- Strong anti-leukemia activity
- Acceptable safety profile
- High response rates in CD123-expressing malignancies
Key Findings
- Monotherapy ORR (AML): 30–40% in some cohorts
- Combination therapy:
- With azacitidine/venetoclax: 60–70% response in relapsed AML
- BPDCN: Clinically meaningful complete responses
Advantages Over Conventional Therapies
- Highly targeted
- Less systemic toxicity
- Better tolerability
- Suitable for older patients or those unfit for intensive chemotherapy
Dosage and Administration
Note: Always refer to the most updated prescribing information.
General Administration
- Administered as an intravenous infusion
- Given in cycles depending on indication and combination protocol
Typical Regimens in Trials
- Once every 21 days
- Combination cycles with azacitidine and venetoclax in AML
- Pre-medication required to prevent infusion reactions
Important Points
- Monitor blood counts
- Dose adjustments may be needed for toxicity
- Prophylactic antimicrobials may be recommended
Side Effects of Pivekimab Sunirine
Common Adverse Effects
- Fatigue
- Nausea
- Fever
- Infusion-related reactions
- Cytopenias (neutropenia, anemia, thrombocytopenia)
Moderate Reactions
- Elevated liver enzymes
- Electrolyte disturbances
- Infections due to immunosuppression
Serious Adverse Effects
- Tumor Lysis Syndrome (TLS)
- Febrile neutropenia
- Severe anemia
- Prolonged bone marrow suppression
Long-term Concerns
- Secondary malignancies (rare but possible)
- Organ toxicity with repeated cycles
Contraindications and Precautions
Contraindications
- Known hypersensitivity
- Severe active infection
- Uncontrolled organ failure
Use With Caution In
- Patients with compromised immunity
- Pregnant or breastfeeding women
- Patients receiving strong CYP-modifying drugs
Monitoring Requirements
- CBC with differential
- Liver and kidney function
- Infection screening
- Infusion reaction monitoring
Drug Interactions
While formal drug interaction data are limited, caution is advised with:
1. Immunosuppressants
May increase infection risk.
2. Strong CYP450 modulators
Could affect metabolism of co-administered agents.
3. Chemotherapy agents
May increase hematologic toxicity.
Patient Counseling Points for Pivekimab Sunirine
Pharmacists should counsel patients on:
- Possible fatigue and weakness
- Increased infection risk
- Importance of monitoring blood counts
- Reporting symptoms like fever, bleeding, or chest discomfort
- Hydration to prevent tumor lysis syndrome
- Infusion reaction symptoms
Also, reinforce that therapy must never be stopped without consulting the oncologist.
Advantages (Why It’s Considered a Breakthrough)
- Highly selective CD123 targeting
- Improved tolerance compared to traditional chemotherapy
- Useful for relapsed/refractory cases
- Lower systemic toxicity
- Good combination synergy with hypomethylating agents and venetoclax
Limitations of Pivekimab Sunirine
- High cost
- Requires infusion center infrastructure
- Limited long-term survival data
- Increased risk of infections
- Not suitable for all leukemia subtypes
Future Prospects –
Researchers are working on:
1. Expanding its use in newly diagnosed AML
With strong early results, it may become a first-line therapy for elderly patients.
2. Combination with other ADCs and immunotherapies
To enhance response rates even further.
3. MRD-focused therapy development
Could help improve remission quality and prevent relapse.
4. Pediatric leukemia trials
Future possibilities remain exciting.
Summary Table – Quick Revision for Exams
| Feature | Details |
| Drug Type | Antibody–Drug Conjugate (ADC) |
| Target | CD123 |
| Payload | Indolinobenzodiazepine (IGN) |
| Primary Use | AML, BPDCN |
| Route | IV infusion |
| Key Benefit | Targeted killing of cancer cells |
| Major Side Effect | Cytopenias, infections |
Conclusion – Why Understanding Pivekimab Sunirine Is Essential
Pivekimab sunirin is a major advance in the treatment of aggressive blood cancers such as AML and BPDCN. Its targeted mechanism, robust clinical data, and promising future applications make it an important molecule for pharmacy students, clinicians, oncologists, and healthcare educators.
As oncology moves toward personalized and targeted therapies, knowledge of drugs like pivekimab sunirin is essential for academic excellence, clinical decision-making, and patient safety.

