Table of Contents
Are you looking for effective treatments for amyloidosis that target the root cause of the disease?
Patisiran amyloidosis therapy is a revolutionary approach to managing hereditary transthyretin-mediated (hATTR) amyloidosis, particularly its debilitating polyneuropathy symptoms. This siRNA interference-based treatment offers a ray of hope by silencing the defective genes responsible for producing toxic proteins. Both healthcare professionals and patients benefit from understanding its role in modern medicine.
What is Patisiran Amyloidosis?
Patisiran amyloidosis refers to the use of patisiran, a pioneering medication, to combat transthyretin amyloidosis (ATTR), a rare condition where misfolded transthyretin proteins form amyloid deposits in tissues. These deposits damage nerves and organs, leading to progressive polyneuropathy, cardiomyopathy, and other complications. In the context of patisiran amyloidosis, the therapy specifically addresses the hereditary forms caused by genetic mutations in the TTR gene.
Pharmacy students should note that patisiran amyloidosis treatment focuses on polyneuropathy, distinguishing it from wild-type ATTR. Patients experience symptoms such as numbness, pain, and weakness, which patisiran amyloidosis interventions aim to halt or reverse. This targeted strategy represents a shift from symptomatic relief to disease-modifying action.
FDA Approval Status
Patisiran received FDA approval in 2018 under the brand name Onpattro for the treatment of polyneuropathy in adults with hereditary ATTR amyloidosis. This rapid approval was granted due to robust Phase 3 trial data demonstrating neurological improvement. Regulatory agencies worldwide, including the European Medicines Agency, followed suit, confirming its safety and efficacy profile.
The approval process highlighted Patisiran’s innovative RNA interference mechanism, earning it orphan drug and breakthrough therapy designations. As of the end of 2025, no major updates have altered its primary polyneuropathy indication. Clinical pharmacists monitor ongoing post-marketing surveillance for expanded use.
Mechanism of Action
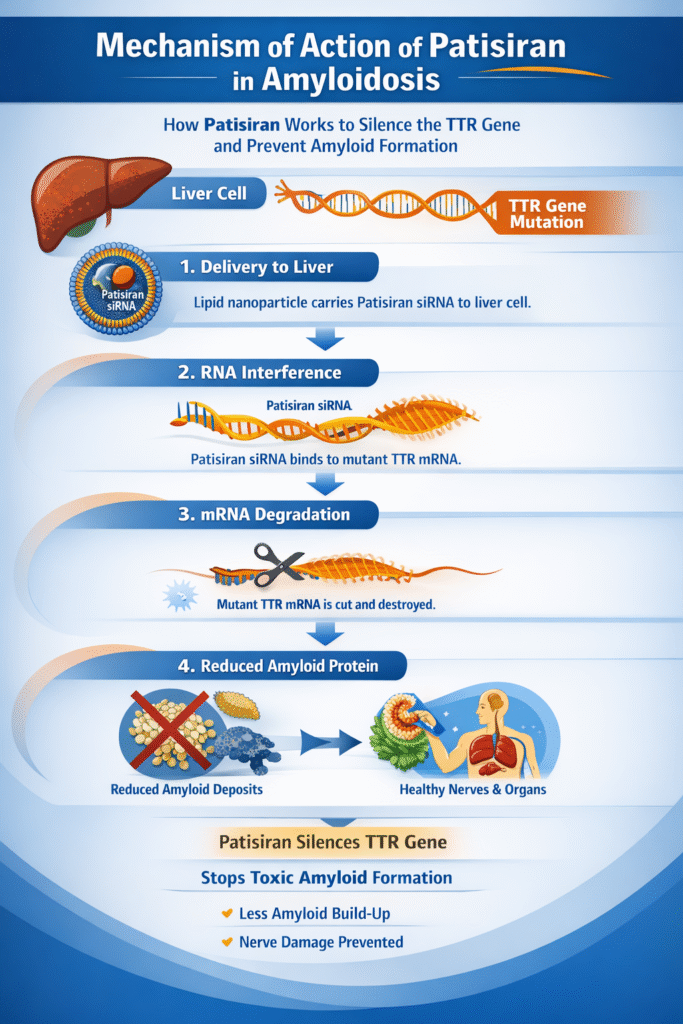
Patisiran utilizes the natural cellular process of RNA interference (RNAi), in which small interfering RNAs (siRNAs) silence specific genes. In simpler terms, in the treatment of amyloidosis, Patisiran delivers siRNAs encapsulated in lipid nanoparticles to the liver, where they bind to the transthyretin (TTR) messenger RNA. This binding triggers the enzymatic degradation of the mRNA, preventing the production of the mutated TTR protein, which misfolds and transforms into amyloid.
Unlike traditional medications, this RNAi approach reduces serum TTR levels by over 80% within weeks, halting amyloid deposition. For patients seeking further clarification, healthcare professionals explain this as “silencing genes at the molecular level.” This liver-specific delivery ensures systemic effects without widespread unwanted side effects.
Indications and Uses
Patisiran is indicated for adults experiencing polyneuropathy due to hereditary ATTR amyloidosis. It slows disease progression, improves neuropathy impairment scores, and enhances quality of life. Although not formally approved, it is used off-label in early-stage cardiomyopathy.
Clinical pharmacists advise on its use in patients with stage 1 or 2 polyneuropathy. The uses of patisiran in amyloidosis do not apply to wild-type ATTR without neuropathy. Dosing is initiated after the diagnosis is confirmed through genetic testing and biopsy.
Dosage and Administration
Administer patisiran as a 0.3 mg/kg intravenous infusion every 3 weeks, diluted in saline. Premedicate with dexamethasone, acetaminophen, and antihistamines to mitigate infusion reactions. Infusion lasts about 45-60 minutes under medical supervision.
Adjust dosage for body weight: for example, 10 mg for a 30-44 kg patient. No oral formulation exists; subcutaneous alternatives are in trials. Pharmacy teams prepare solutions fresh, discarding unused portions after 24 hours refrigerated.
- Preparation steps: Calculate dose by weight, thaw frozen vials in fridge, mix gently.
- Monitoring: Vital signs during first infusions; train home administration if approved.
Pharmacokinetics and Dynamics
Patisiran nanoparticles are rapidly absorbed into liver cells after infusion, achieving the maximum reduction in TTR levels within 13 days. The lipid components have a half-life of 4-6 days, and the siRNA is eliminated through urinary and metabolic pathways. After multiple doses, a sustained reduction of 81-96% in TTR levels is observed.
Pharmacodynamically, the reduction in TTR levels is directly linked to the stabilization of neuropathy. There is no accumulation of the drug in the body with dosing every three weeks. No adjustments are required for special patient populations, such as those with renal impairment, which helps in meeting the needs of diverse patients.
Clinical Trial Evidence
The pivotal APOLLO trial (Phase 3) enrolled 225 hATTR patients, who were randomized to patisiran or placebo. The primary endpoint was met: 56% less neuropathy progression compared to a 0.9-point worsening in the placebo group (p<0.001). Secondary benefits included improvements in walking speed, quality of life, and autonomic function.
Five-year open-label extensions confirmed sustained efficacy, with many patients remaining neurologically stable. Real-world data mirrors the trial results, emphasizing the importance of early intervention. Patisiran’s amyloidosis outcomes are superior to historical stabilizers like tafamidis in neuropathy-focused metrics.
Benefits in Amyloidosis
Patisiran treatment for amyloidosis halts the progression of polyneuropathy in 50-60% of patients and reverses symptoms in one-third of patients. A 14-point improvement in the Norfolk quality of life score indicates functional benefits. Stabilization of the cardiovascular system emerges as an additional benefit, leading to reduced hospitalizations.
Patients report less pain and improved mobility, granting them independence for daily activities. For pharmacists, the delay in disability improves cost-effectiveness in the long run. The benefits of Patisiran for amyloidosis extend to family counseling regarding genetic risks.
Side Effects and Safety
Common side effects include infusion-related reactions (19%), peripheral edema (18%), and nausea (12%). Most resolve without discontinuation. Serious events like thrombocytopenia occur rarely (<1%).
Long-term safety shows no new signals over five years. Monitoring includes platelet counts and liver enzymes quarterly. Patisiran amyloidosis profiles favor its risk-benefit in progressive disease.
Warnings and Precautions
Use with caution in severe hepatic impairment; monitor vitamin A levels due to TTR transport role. Infusion reactions require prompt intervention with steroids. Contraception is required in women of childbearing potential; breastfeeding data is limited.
Contraindications: Hypersensitivity to the components. Precautions for dialysis patients include timing the infusion according to post-dialysis sessions. Clinical pharmacists document a history of hypersensitivity prior to infusion.
Drug Interactions
Patisiran amyloidosis therapy interacts minimally due to hepatic targeting. Cyclosporine mildly elevates exposure; monitor with concurrent immunosuppressants. No major cytochrome P450 effects, simplifying polypharmacy.
Acid reducers like PPIs do not impact RNAi efficacy. Rifampin may reduce TTR silencing slightly. Advise spacing from hepatotoxins.
| Drug Class | Interaction Level | Recommendation |
| Immunosuppressants | Moderate | Monitor TTR levels |
| CYP Inducers | Low | No dose change |
| Antacids | None | Safe co-use |
| Hepatotoxins | Moderate | Liver function tests |
Comparison with Other Treatments
Patisiran is more effective at halting disease progression than stabilizers such as diflunisal or doxycycline. Compared to tafamidis (an oral stabilizer), patisiran provides more profound suppression of TTR (80% versus 36%), but it requires infusion.
Inotersen (an antisense oligonucleotide) has similar efficacy, but it carries a risk of thrombocytopenia. Vutrisiran, a subcutaneous RNAi drug, offers similar benefits with administration every three months.
| Treatment | Route | TTR Reduction | Neuropathy Halt Rate | Key Drawback |
| Patisiran | IV q3w | 80-90% | 56% | Infusions |
| Tafamidis | Oral daily | 36% | 45% | Less potent |
| Inotersen | SC weekly | 70-80% | 50% | Platelet drop |
| Vutrisiran | SC q3m | 85% | 55% | Injection site |
Patient Counseling Points
Pharmacy-focused counseling emphasizes pre-treatment adherence to avoid adverse reactions. Stress the third-week schedule; missed doses increase the risk of TTR recurrence. Educate about signs of progression: increasing numbness indicates the need for evaluation.
Avoiding dietary vitamin A prevents toxicity. Home infusion training includes storage at -20°C. Patisiran amyloidosis success depends on adherence; track via apps.
• Recognize infusion signs: chills, flushing—stop and call the provider.
• Genetic counseling for family members.
• Report new weakness immediately.
Future Scope and Research
Cardiomyopathy (post-HELIOS-B) and pediatric applications of patisiran in amyloidosis are currently under investigation. Subcutaneous formulations are intended for convenience. Combination with stabilizers is being explored for synergy.
Gene editing complements long-term RNAi. Real-world registries track durability for over 10 years. Patisiran in amyloidosis evolves with precision medicine advancements.
Conclusion
Patisiran amyloidosis therapy offers a definitive positive approach to effective amyloidosis treatment, transforming patients’ lives through precise gene silencing and neuropathy control.. Early adoption maximizes outcomes for patients and guides pharmacy excellence.