Table of Contents
Introduction
The year 2025 marked a watershed moment in endocrinology with the FDA’s approval of Paltusotin (brand name: Palsonify) on September 25, 2025. This milestone marks the beginning of a once-daily oral therapy for adults with acromegaly who have had an inadequate response to surgery or for whom surgery is not an option.
The Paltusotine FDA approval represents a major shift from traditional injectable treatments, providing patients with a more convenient and effective solution. This advancement is a ray of hope for those struggling with the chronic and often debilitating symptoms of acromegaly.
In this blog, we will explore what Paltusotin is, how it works, its clinical benefits, dosing information, side effects, and why this FDA approval is being called the most significant endocrinology advancement of the decade.
What Is Paltusotine (Palsonify)?
Paltusotin, marketed as Palsonify, is a selective, oral, non-peptide somatostatin receptor type 2 (SST2) agonist developed by Crinetics Pharmaceuticals. It is designed to mimic the action of somatostatin, a natural hormone that helps the pituitary gland release growth hormone (GH).
In patients with acromegaly, excessive secretion of growth hormone causes abnormal growth of body tissues, leading to symptoms such as enlarged hands and feet, joint pain, headaches, and facial changes.
Prior to Paltusotine FDA approval, most acromegaly treatments required monthly injections such as octreotide LAR or lanreotide depot, which can be painful and inconvenient. Paltusotin changes that story completely – offering a once-daily oral tablet that can maintain growth hormone control without injections.
Paltusotine FDA Approval: Key Details
| Parameter | Information |
| Drug Name | Paltusotine |
| Brand Name | Palsonify |
| Approval Date | September 25, 2025 |
| Approved By | U.S. Food and Drug Administration (FDA) |
| Developer | Crinetics Pharmaceuticals |
| Indication | Treatment of adults with acromegaly who have inadequate response to surgery or are not surgical candidates |
| Dosage Form | Oral tablet |
| Class | Somatostatin receptor agonist (SST2 selective) |
The FDA approval of paltusotine followed strong evidence from Phase 3 clinical trials (PATHFNDR-1 and PATHFNDR-2), which demonstrated that the oral therapy maintained growth hormone and insulin-like growth factor-1 (IGF-1) levels in patients who switched from injectable treatments.
This approval marks the first oral, nonpeptide somatostatin analog ever approved for acromegaly in the U.S.
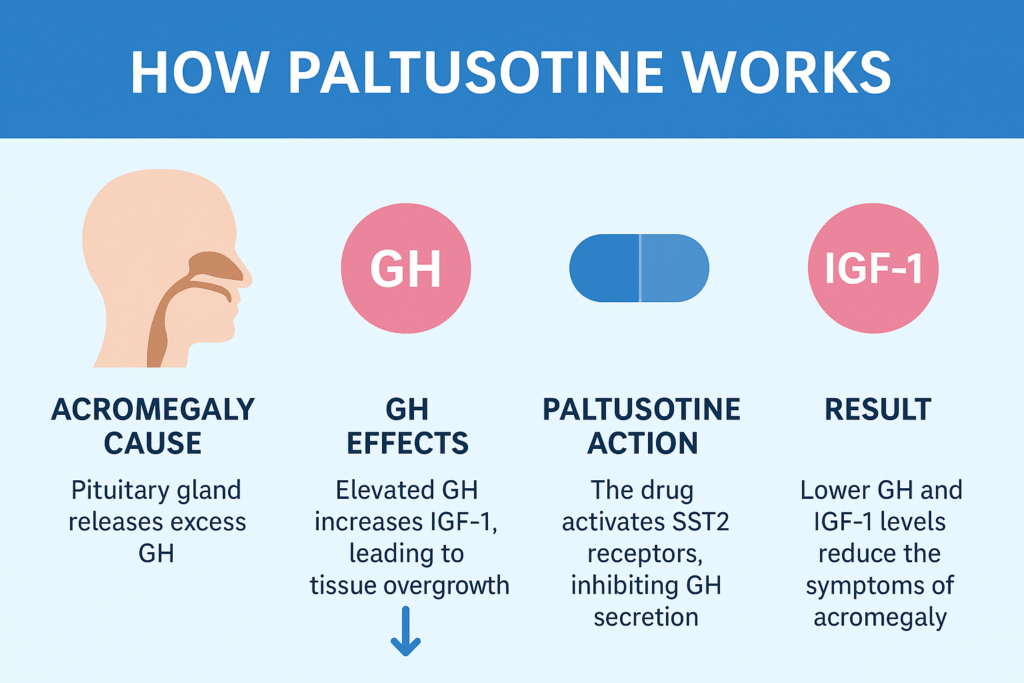
How Paltusotine Works: Mechanism of Action
Paltusotine acts by binding to somatostatin receptor subtype 2 (SST2) on pituitary cells, which suppresses the release of growth hormone (GH).
Here’s a simplified mechanism:
- Acromegaly Cause: The pituitary gland releases excess GH.
- GH Effects: Elevated GH increases IGF-1 levels, leading to tissue overgrowth.
- Paltusotine Action: The drug activates SST2 receptors, inhibiting GH secretion.
- Result: Lower GH and IGF-1 levels reduce the symptoms and complications of acromegaly.
Unlike older injectable analogs that are peptide-based and require depot injections, paltusotine’s nonpeptide design allows it to be absorbed orally and maintain stable plasma concentrations, mimicking continuous somatostatin suppression.
Clinical Trial Evidence Supporting Paltusotine FDA Approval
The paltusotine FDA approval was primarily based on the results of two pivotal Phase 3 studies — PATHFNDR-1 and PATHFNDR-2.
PATHFNDR-1 Study
- Design: Randomized, double-blind, placebo-controlled.
- Participants: Adults with acromegaly previously managed with injectables.
- Objective: Assess maintenance of IGF-1 control after switching to oral paltusotine.
- Results:
- 83% of participants maintained biochemical control (normal IGF-1).
- Comparable GH suppression to prior injectable therapy.
- No significant rebound or loss of efficacy.
PATHFNDR-2 Study
- Design: Evaluated patients naive to medical therapy or post-surgery with residual disease.
- Results:
- Paltusotine significantly reduced IGF-1 from baseline.
- Improved symptom control and patient quality of life.
- Fewer injection-related side effects and better treatment adherence.
These studies demonstrated that oral paltusotine was as effective as injectable octreotide or lanreotide, paving the way for its FDA approval in 2025.
Advantages of Paltusotine Over Traditional Treatments
| Feature | Traditional Injectable Therapies | Paltusotine (Palsonify) |
| Route of Administration | Monthly or biweekly injections | Once-daily oral tablet |
| Convenience | Requires healthcare visits | Home-based self-administration |
| Pain/Discomfort | Injection site pain common | No injection-related issues |
| Efficacy | Effective, but variable absorption | Comparable biochemical control |
| Adherence | Often low due to injections | Improved due to oral dosing |
This oral alternative is expected to increase treatment adherence, minimize clinic visits, and improve the overall quality of life for patients with acromegaly.
Dosage and Administration
Paltusotine is available as oral tablets in varying strengths.
- Starting Dose: Typically 10 mg once daily.
- Maintenance Dose: 10–40 mg daily based on IGF-1 levels and clinical response.
- Administration Tips:
- Take at the same time each day.
- Should be taken on an empty stomach (at least 1 hour before or 2 hours after a meal).
- Swallow whole with water; do not crush or chew.
Dosing adjustments are made every 4 to 8 weeks depending on laboratory and clinical evaluations.
Side Effects of Paltusotine
Like any medication, Paltusotine (Palsonify) may cause side effects. However, most are mild to moderate and tend to improve with time.
Common Side Effects
- Nausea or upset stomach
- Abdominal pain
- Diarrhea or constipation
- Headache
- Fatigue
- Mild glucose intolerance
Serious but Rare Side Effects
- Bradycardia (slow heart rate)
- Gallstones
- Hypoglycemia or hyperglycemia
- Hypothyroidism
Patients should report any unusual symptoms to their healthcare provider promptly.
Clinical trials showed that paltusotine had a favorable safety profile, with fewer gastrointestinal and injection-site issues compared to traditional therapies.
Paltusotine and Quality of Life
The paltusotine FDA approval goes beyond clinical efficacy—it represents an improvement in patient convenience, comfort, and adherence.
Many acromegaly patients have expressed frustration with the inconvenience and pain of injectable treatments. The ability to take a simple daily oral pill instead of monthly injections significantly enhances treatment satisfaction.
Moreover, paltusotine’s oral formulation reduces:
- Clinic visits
- Injection anxiety
- Travel costs and time off work
This shift supports patient independence and long-term disease management.
Cost and Accessibility
Following the paltusotine FDA approval, Crinetics Pharmaceuticals announced its plans to launch Palsonify in the U.S. market in late 2025.
While pricing details are still under review, experts expect the cost to be comparable to existing injectable analogs, with potential patient assistance programs to ensure accessibility.
Insurance coverage is expected to expand quickly, given its FDA-approved indication and superior patient convenience profile.
Future Implications of Paltusotine FDA Approval
The paltusotine FDA approval has implications far beyond acromegaly treatment:
- Sets precedent for oral nonpeptide hormone therapies.
- Encourages development of next-generation somatostatin analogs for other hormonal disorders (e.g., neuroendocrine tumors).
- Demonstrates the viability of oral bioavailability for complex peptide analogs.
Crinetics is also exploring paltusotine’s potential in treating carcinoid syndrome and other GH-related disorders, marking the beginning of a new era in endocrine therapy.
Conclusion
The Paltusotine FDA Approval marks a pivotal moment in the evolution of acromegaly treatment. As the first oral somatostatin receptor agonist to be taken once daily, Paltusotin (Palsonify) transforms how this chronic hormonal disorder is managed.
Patients can now control their growth hormone levels without injections, improving adherence, convenience, and overall quality of life.
With its robust efficacy, favorable safety, and patient-centered design, Paltusotin is not just a drug – it is a revolution in endocrine therapy.
The future of acromegaly management is here, and it starts with Paltusotin (Palsonify) – a small pill with a big impact.

