Table of Contents
The global burden of obesity and type 2 diabetes continues to rise at an alarming rate. As healthcare systems search for innovative and patient-friendly therapies, oral GLP-1 receptor agonists have gained significant attention. Among the most anticipated therapies in this category is orforglipron. The discussion surrounding orforglipron approval fda represents a pivotal moment in metabolic medicine.
Orforglipron is an investigational oral small-molecule GLP-1 receptor agonist developed by Eli Lilly and Company. Unlike injectable GLP-1 drugs, this therapy is designed for convenient oral administration without peptide-based delivery constraints. The potential impact of orforglipron approval fda extends beyond convenience—it may reshape treatment strategies for millions of patients worldwide.
In this comprehensive guide, we will explore:
- Mechanism of Action (MOA)
- Clinical trial evidence
- Side effects and safety profile
- Drug interactions
- Regulatory pathway and FDA considerations
- Future implications for obesity and diabetes care
Understanding Orforglipron
Orforglipron is a non-peptide, small-molecule GLP-1 receptor agonist. Unlike traditional GLP-1 drugs such as semaglutide, which are peptide-based and require injections or special absorption techniques, orforglipron is engineered to be taken orally without strict fasting requirements.
The anticipation around orforglipron approval fda stems from its potential to combine:
- Strong glycemic control
- Significant weight reduction
- Oral convenience
- Improved adherence
If granted regulatory clearance, orforglipron approval fda could represent a major advancement in metabolic therapeutics.
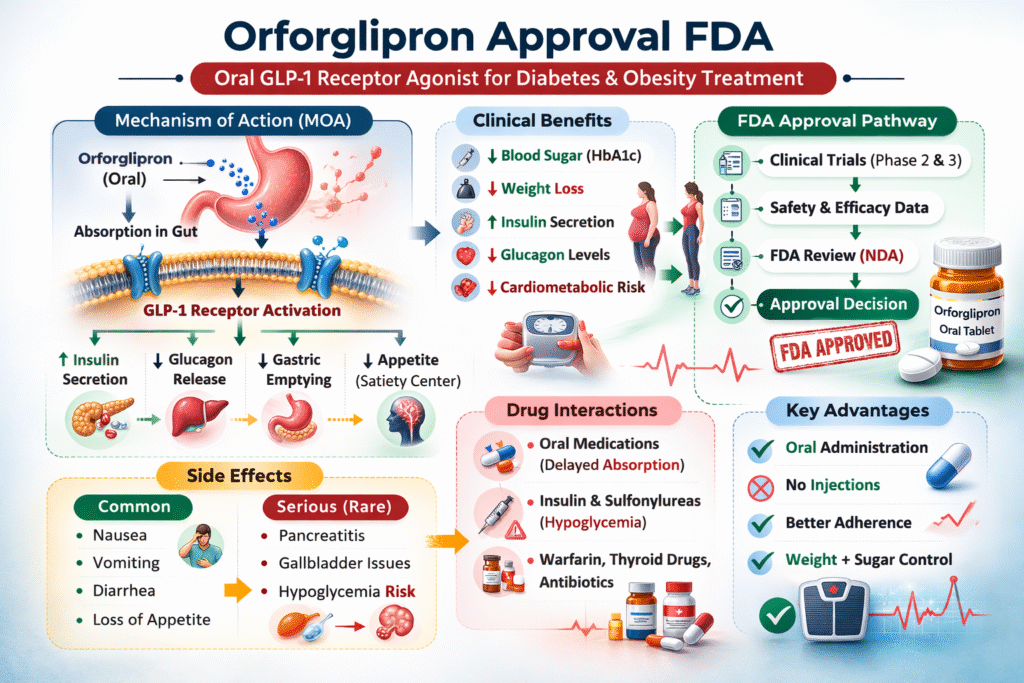
Mechanism of Action (MOA)
GLP-1 Receptor Activation
Orforglipron works by activating the glucagon-like peptide-1 (GLP-1) receptor. GLP-1 is an incretin hormone that:
- Stimulates insulin secretion (glucose-dependent)
- Suppresses glucagon release
- Delays gastric emptying
- Promotes satiety
Through selective GLP-1 receptor activation, orforglipron improves postprandial glucose control while reducing caloric intake.
The scientific innovation behind orforglipron approval fda lies in its non-peptide structure. It binds to the GLP-1 receptor in a way that mimics endogenous hormone activity but allows oral bioavailability.
Metabolic Effects
Following receptor activation:
- Pancreatic beta cells increase insulin secretion
- Alpha cells reduce glucagon output
- Gastric emptying slows
- Hypothalamic appetite pathways are modulated
This integrated metabolic regulation explains why expectations for orforglipron approval fda are high in both diabetes and obesity management.
Clinical Trial Evidence
Phase 2 and Phase 3 Data
Clinical studies evaluating orforglipron demonstrated:
- Significant HbA1c reduction
- Dose-dependent weight loss
- Improvements in cardiometabolic markers
In multiple trials, patients experienced meaningful reductions in body weight, rivaling injectable GLP-1 agents. These findings strongly support the case for orforglipron approval fda.
Weight Reduction Outcomes
Participants receiving higher doses showed:
- Double-digit percentage weight loss
- Reduced waist circumference
- Improved lipid profiles
The obesity epidemic creates a compelling case for orforglipron approval fda, particularly as oral administration may improve long-term compliance.
FDA Approval Pathway
Regulatory Considerations
The United States Food and Drug Administration (U.S. Food and Drug Administration) evaluates drugs based on:
- Safety
- Efficacy
- Manufacturing quality
- Risk-benefit ratio
The review process includes:
- New Drug Application (NDA) submission
- Advisory committee review (if required)
- Labeling negotiations
- Post-marketing surveillance commitments
The progress toward orforglipron approval fda depends on long-term cardiovascular outcomes and safety data. Orforglipron a once-daily oral GLP-1 receptor agonist for weight loss and type 2 diabetes developed by Eli Lilly, is not yet FDA approved but is expected to receive a regulatory decision by April 10, 2026.
Potential Indications
If regulatory approval is granted, anticipated indications include:
- Type 2 diabetes mellitus
- Chronic weight management
- Possibly cardiometabolic risk reduction
Each indication strengthens the clinical relevance of orforglipron approval fda.
Side Effects Profile
As with all GLP-1 receptor agonists, gastrointestinal effects are the most commonly reported.
Common Side Effects
- Nausea
- Vomiting
- Diarrhea
- Constipation
- Reduced appetite
These effects are generally dose-dependent and often improve with continued therapy.
The safety evaluation plays a central role in determining orforglipron approval fda, especially regarding tolerability and discontinuation rates.
Serious Adverse Events
Potential but less common risks include:
- Pancreatitis
- Gallbladder disease
- Severe gastrointestinal intolerance
Regulatory authorities carefully assess these risks before finalizing orforglipron approval fda.
Drug Interactions
Because orforglipron slows gastric emptying, it may affect absorption of:
- Oral antibiotics
- Thyroid medications
- Oral contraceptives
- Warfarin
Caution is advised when co-administered with insulin or sulfonylureas due to hypoglycemia risk.
Comprehensive interaction data is crucial for orforglipron approval fda, ensuring prescribers have clear guidance.
Cardiovascular Implications
GLP-1 receptor agonists have demonstrated cardiovascular benefits in prior outcome trials. Regulators may require cardiovascular outcome trials (CVOT) to confirm safety.
If long-term data confirms benefit or neutrality, it will strongly support orforglipron approval fda for broader indications.
Advantages Over Injectable GLP-1 Drugs
Oral administration provides:
- Increased convenience
- Better patient acceptance
- Reduced needle anxiety
- Potentially improved adherence
These factors significantly enhance the value proposition of orforglipron approval fda in real-world settings.
Market and Public Health Impact
The global diabetes and obesity market is expanding rapidly. Oral GLP-1 therapy could:
- Expand access to early treatment
- Reduce complications
- Improve long-term metabolic outcomes
The healthcare impact of orforglipron approval fda may extend well beyond individual patient benefits.
Safety Monitoring Post-Approval
If approved, post-marketing surveillance will monitor:
- Rare adverse effects
- Long-term cardiovascular outcomes
- Real-world tolerability
Ongoing pharmacovigilance remains essential even after orforglipron approval fda.
Future Research Directions
Potential areas of investigation include:
- Combination with SGLT2 inhibitors
- Use in prediabetes
- NAFLD/NASH treatment
- Cardiovascular risk reduction
Such expansions would further amplify the significance of orforglipron approval fda.
Conclusion
The journey toward orforglipron approval fda represents a defining moment in metabolic therapeutics. As an oral non-peptide GLP-1 receptor agonist, orforglipron offers:
- Strong glycemic control
- Significant weight loss
- Improved patient convenience
- Potential cardiometabolic benefits
The final regulatory decision regarding orforglipron approval fda will depend on comprehensive safety, efficacy, and cardiovascular outcome data. However, current evidence suggests that this therapy could transform the management of obesity and type 2 diabetes.
With continued clinical development and regulatory evaluation, orforglipron approval fda may soon become one of the most impactful milestones in modern endocrinology.

