Table of Contents
Introduction
Motion sickness, a common yet debilitating condition, has long plagued travelers of all kinds. From bumpy car rides to turbulent flights and rocky boat trips, the disorienting symptoms of nausea, dizziness, and vomiting can transform an anticipated journey into an ordeal. For years, the available treatments have been limited, often offering incomplete relief or undesirable side effects. This landscape, however, is now set to change dramatically with the groundbreaking Nereus FDA approval.
The recent Nereus FDA approval marks a significant milestone in medical science, introducing a novel therapeutic option that promises to redefine how motion sickness is managed. This comprehensive blog post will delve into the intricacies of Nereus, exploring its mechanism of action, the journey to its Nereus FDA approval, its side effects, drug interactions, administration, precautions, and ultimately, its potential impact on millions worldwide. The Nereus FDA approval is not just another regulatory decision; it represents a new hope for those who have long suffered in silence.
Understanding Motion Sickness: The Unmet Need
Before we delve into the specifics of the Nereus FDA approval, it’s crucial to understand the condition it aims to alleviate. Motion sickness arises from a conflict between the sensory inputs our brain receives. Our eyes might see a static environment inside a car, but our inner ear (vestibular system) detects motion, creating a sensory mismatch. This confusion triggers a cascade of physiological responses, leading to the familiar symptoms.
For many, current over-the-counter options like dimenhydrinate (Dramamine) and meclizine, or prescription scopolamine patches, come with limitations such as drowsiness, dry mouth, or only partial efficacy. The long-awaited **Nereus FDA approval addresses this critical unmet need, offering a potentially more effective and tolerable treatment. The journey to the Nereus FDA approvalwas driven by the desire to provide a superior solution.
Nereus: Mechanism of Action (MOA)
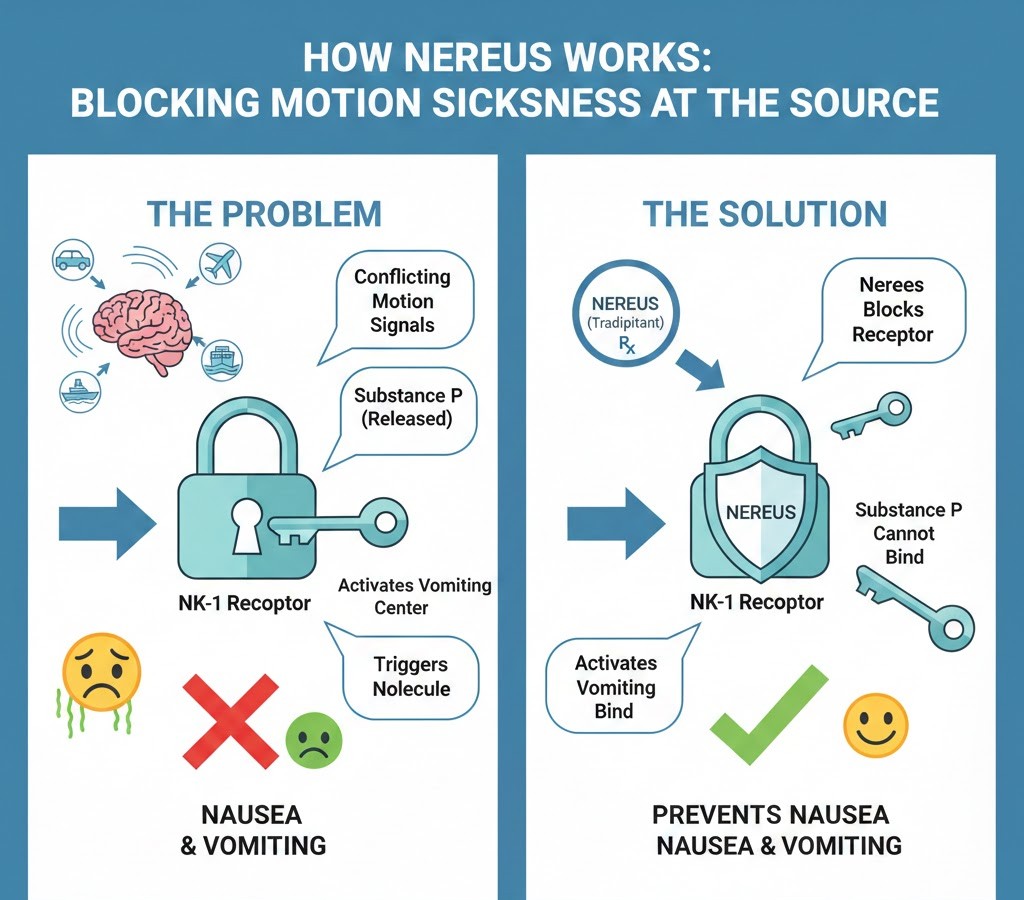
The scientific brilliance behind Nereus lies in its innovative mechanism of action. Nereus, with its active ingredient tradipitant, is a neurokinin-1 (NK-1) receptor antagonist. To understand what this means, we need a brief foray into neurobiology. Substance P is a neuropeptide that plays a crucial role in various physiological processes, including pain transmission, inflammation, and critically, the emetic (vomiting) reflex. When the brain receives conflicting sensory signals leading to motion sickness, Substance P is released in certain areas, particularly the nucleus tractus solitarius and the area postrema, which are key regions involved in nausea and vomiting.
NK-1 receptors are the specific docking sites for Substance P. By acting as an antagonist, tradipitant in Nereus effectively blocks Substance P from binding to these NK-1 receptors. This blockage disrupts the signaling pathway that leads to nausea and vomiting associated with motion sickness. Unlike older antihistamine-based treatments that primarily induce sedation, Nereus targets a specific neurochemical pathway directly involved in the emetic reflex, offering a more precise and potentially more effective antiemetic effect without the same level of sedative side effects. This targeted approach is a key differentiator highlighted by the Nereus FDA approval. The unique MOA of Nereus was a central aspect considered during its Nereus FDA approval.
The Journey to Nereus FDA Approval
The path to Nereus FDA approval was a rigorous and lengthy process, typical for novel drugs. It involved extensive preclinical research, multiple phases of clinical trials, and thorough review by the Food and Drug Administration (FDA).
Preclinical Studies: Early laboratory and animal studies were conducted to understand Nereus’s pharmacology, toxicology, and initial efficacy in models of nausea and vomiting. These studies laid the groundwork for human trials.
Phase 1 Clinical Trials: These trials involved a small number of healthy volunteers to assess Nereus’s safety, tolerability, pharmacokinetics (how the body absorbs, distributes, metabolizes, and excretes the drug), and pharmacodynamics (what the drug does to the body).
Phase 2 Clinical Trials: Larger groups of patients experiencing motion sickness were enrolled to evaluate Nereus’s efficacy, determine optimal dosing, and further assess safety. These trials provided initial evidence of Nereus’s effectiveness in a real-world setting.
Phase 3 Clinical Trials: The pivotal trials for Nereus FDA approval involved hundreds, if not thousands, of patients across multiple centers. These large-scale, placebo-controlled studies confirmed Nereus’s efficacy, safety profile, and tolerability compared to existing treatments or placebo. The results demonstrated a statistically significant reduction in the incidence and severity of motion sickness symptoms, including nausea, vomiting, and dizziness, across various simulated and real-world motion environments (e.g., ferry rides, virtual reality motion simulators). The compelling data from these trials were instrumental in securing the Nereus FDA approval.
FDA Review: Following the submission of a New Drug Application (NDA) containing all the preclinical and clinical data, the FDA meticulously reviewed every aspect of Nereus. This included evaluating manufacturing processes, drug quality, and the overall risk-benefit profile. After a comprehensive assessment, the FDA concluded that Nereus was safe and effective for its intended use, leading to the landmark Nereus FDA approval on December 30, 2025. This decision signifies a major advancement in the treatment of motion sickness. The thoroughness of the FDA review underscored the importance of the Nereus FDA approval.
Side Effects
While the Nereus FDA approval brings a valuable new treatment option, it’s essential for patients and healthcare providers to be aware of its potential side effects. Like all medications, Nereus is associated with certain adverse reactions, though clinical trials demonstrated generally good tolerability.
Commonly reported side effects during trials for Nereus FDA approval included:
Headache: This was one of the most frequently observed side effects, typically mild to moderate in intensity.
Dizziness: While Nereus aims to reduce motion sickness-related dizziness, some patients reported general dizziness as a side effect.
Fatigue: A feeling of tiredness or low energy was also noted in some individuals.
Dry Mouth: Though less common than with some older antihistamines, dry mouth was reported by a subset of patients.
Constipation: Gastrointestinal disturbances, including constipation, were occasionally observed.
Less common but potentially more serious side effects may include:
Allergic Reactions: As with any medication, there is a risk of allergic reactions, ranging from rash and itching to more severe anaphylaxis. Patients should seek immediate medical attention if they experience symptoms of a severe allergic reaction.
Liver Enzyme Elevations: In some clinical trials, transient and asymptomatic elevations in liver enzymes were observed. Regular monitoring of liver function may be recommended for certain patients.
It is crucial for patients to discuss any concerns about side effects with their healthcare provider. The comprehensive data leading to Nereus FDA approval includes a detailed safety profile that physicians can use to guide treatment decisions. The careful evaluation of side effects was paramount to the Nereus FDA approval.
Drug Interactions
Understanding potential drug interactions is vital to ensure the safe and effective use of Nereus. As Nereus (tradipitant) is metabolized in the liver, primarily through the cytochrome P450 (CYP) enzyme system, it may interact with other medications that affect these enzymes.
Key potential drug interactions to be aware of include:
CYP3A4 Inhibitors: Strong inhibitors of CYP3A4, such as ketoconazole (antifungal), clarithromycin (antibiotic), and ritonavir (antiretroviral), can increase the blood levels of Nereus, potentially leading to an increased risk of side effects. Dosage adjustments or careful monitoring may be necessary.
CYP3A4 Inducers: Conversely, strong inducers of CYP3A4, such as rifampin (antibiotic), carbamazepine (antiepileptic), and St. John’s Wort, can decrease the blood levels of Nereus, potentially reducing its efficacy.
Other Medications that Cause Dizziness or Drowsiness: Concurrent use of Nereus with other central nervous system depressants, including alcohol, sedatives, tranquilizers, or other motion sickness medications, may exacerbate dizziness or drowsiness.
QT-Prolonging Drugs: While not a primary concern, caution should be exercised when Nereus is used with other medications known to prolong the QT interval, as this could theoretically increase the risk of certain cardiac arrhythmias.
Patients should always inform their doctor and pharmacist about all prescription, over-the-counter, and herbal medications they are taking before starting Nereus. This information is critical for healthcare providers to assess potential interactions and ensure safe prescribing practices, a key consideration during the Nereus FDA approval process. The vigilance around drug interactions contributes to the overall safety profile of Nereus following its Nereus FDA approval.
Administration and Dosage
The Nereus FDA approval specifies clear guidelines for its administration and dosage. Nereus is typically administered orally, in tablet form.
Recommended Dosage:The standard recommended dosage for adults for the prevention of motion sickness is [specific dosage amount and frequency, e.g., one 20 mg tablet taken orally approximately 1 hour before anticipated travel].
Timing: For optimal effectiveness, Nereus should be taken prior to exposure to motion. Taking it too late may reduce its preventive benefits.
With or Without Food: Nereus can generally be taken with or without food, though patients should follow specific instructions from their healthcare provider or the product labeling.
Special Populations: Dosage adjustments may be considered for individuals with severe renal or hepatic impairment, or for the elderly, although specific recommendations are usually detailed in the official prescribing information resulting from the Nereus FDA approval.
Patients should adhere strictly to the prescribed dosage and not exceed the recommended amount. If a dose is missed, it should be taken as soon as remembered, unless it is almost time for the next dose. Double dosing to compensate for a missed dose is not recommended. The precise administration guidelines are a crucial part of the Nereus FDA approval.
Precautions
Several precautions should be considered before and during treatment with Nereus, as outlined by the comprehensive review that led to Nereus FDA approval.
Hypersensitivity: Nereus is contraindicated in patients with known hypersensitivity to tradipitant or any component of the formulation.
Hepatic Impairment: Patients with moderate to severe liver impairment may require dosage adjustments or closer monitoring due to altered drug metabolism.
Renal Impairment: Similar considerations apply to patients with severe kidney impairment.
Pregnancy and Lactation: The safety of Nereus in pregnant or breastfeeding women has not been fully established. It should only be used if the potential benefit outweighs the potential risk to the fetus or infant. Women of childbearing potential should discuss effective contraception with their doctor.
Pediatric Use: The Nereus FDA approval is specifically for adults. The safety and efficacy of Nereus in pediatric populations have not yet been established.
Driving and Operating Machinery: While Nereus is designed to be less sedating than some older medications, some individuals may still experience dizziness or fatigue. Patients should exercise caution when driving or operating heavy machinery until they know how Nereus affects them.
Underlying Medical Conditions: Patients with pre-existing cardiac conditions or other significant medical issues should discuss these with their healthcare provider before starting Nereus.
Always consult with a healthcare professional to determine if Nereus is appropriate for your specific circumstances, especially considering the comprehensive information available post-Nereus FDA approval.The precautions serve to maximize patient safety following the Nereus FDA approval.
Conclusion: A New Era in Motion Sickness Management
The Nereus FDA approvalon December 30, 2025, represents a transformative moment in the management of motion sickness. For countless individuals whose lives have been limited by the fear and discomfort of travel, Nereus offers a beacon of hope. Its targeted mechanism of action, selectively blocking NK-1 receptors, promises a more effective and potentially better-tolerated alternative to existing treatments. The rigorous journey through preclinical and clinical trials, culminating in the FDA’s thorough review, underscores the robust scientific evidence supporting Nereus’s safety and efficacy.
While no medication is without its side effects or potential interactions, the comprehensive information now available provides healthcare professionals with the tools to prescribe Nereus responsibly and effectively. This new option is poised to significantly improve the quality of life for millions, enabling them to enjoy travel, whether for leisure or necessity, without the dread of debilitating symptoms. The Nereus FDA approval is a testament to ongoing innovation in pharmaceuticals and a clear victory for patient well-being. This significant Nereus FDA approval will undoubtedly change travel experiences globally. The impact of the Nereus FDA approval will be felt for years to come. The Nereus FDA approval solidifies its place as a groundbreaking treatment.

