Table of Contents
Introduction
The navepegritide FDA approval marks a historic milestone in pediatric endocrinology, especially for the treatment of rare growth disorders such as achondroplasia. In recent years, advances in peptide therapeutics have transformed how clinicians approach hormone-related conditions, and navepegritide represents one of the most promising breakthroughs.
On February 27, 2026, the navepegritide FDA approval was granted under the accelerated approval pathway for Yuviwel (navepegritide). This approval specifically targets children aged 2 years and older with achondroplasia who have open growth plates. This development is particularly significant because it introduces the first once-weekly, subcutaneous C-type natriuretic peptide (CNP) analogue designed to increase height velocity.
The importance of the navepegritide FDA approval extends beyond convenience—it directly addresses unmet medical needs in patients suffering from impaired bone growth due to genetic mutations affecting endochondral ossification.
What is Navepegritide (Yuviwel)?
Navepegritide, marketed as Yuviwel, is a long-acting analog of C-type natriuretic peptide (CNP), a naturally occurring peptide that plays a critical role in bone growth regulation.
The navepegritide introduces a therapy that mimics and enhances the biological activity of endogenous CNP, thereby promoting normal skeletal development in children with achondroplasia.
Key Characteristics
- Long-acting peptide analog
- Once-weekly subcutaneous administration
- Targets growth plate physiology
- Specifically designed for pediatric use
The navepegritide FDA approval highlights its innovation as a disease-modifying therapy rather than just a symptomatic treatment.
Understanding Achondroplasia
Achondroplasia is the most common form of disproportionate short stature caused by mutations in the FGFR3 gene. This mutation leads to abnormal cartilage formation and impaired bone growth.
Clinical Features
- Short limbs relative to trunk
- Delayed motor development
- Spinal abnormalities
- Reduced height velocity
The navepegritide FDA approval provides a targeted approach to counteract the molecular mechanisms underlying this condition.
Navepegritide FDA Approval: Regulatory Breakthrough
The navepegritide FDA approval was granted through the accelerated approval pathway, emphasizing the urgent need for effective therapies in rare pediatric conditions.
Approval Highlights
- Brand Name: Yuviwel
- Approval Date: February 27, 2026
- Indication: Achondroplasia in children ≥2 years with open growth plates
- Dosage Form: Subcutaneous injection
- Frequency: Once weekly
This navepegritide FDA approval is groundbreaking because it introduces the first weekly CNP analog, significantly reducing treatment burden compared to daily therapies.
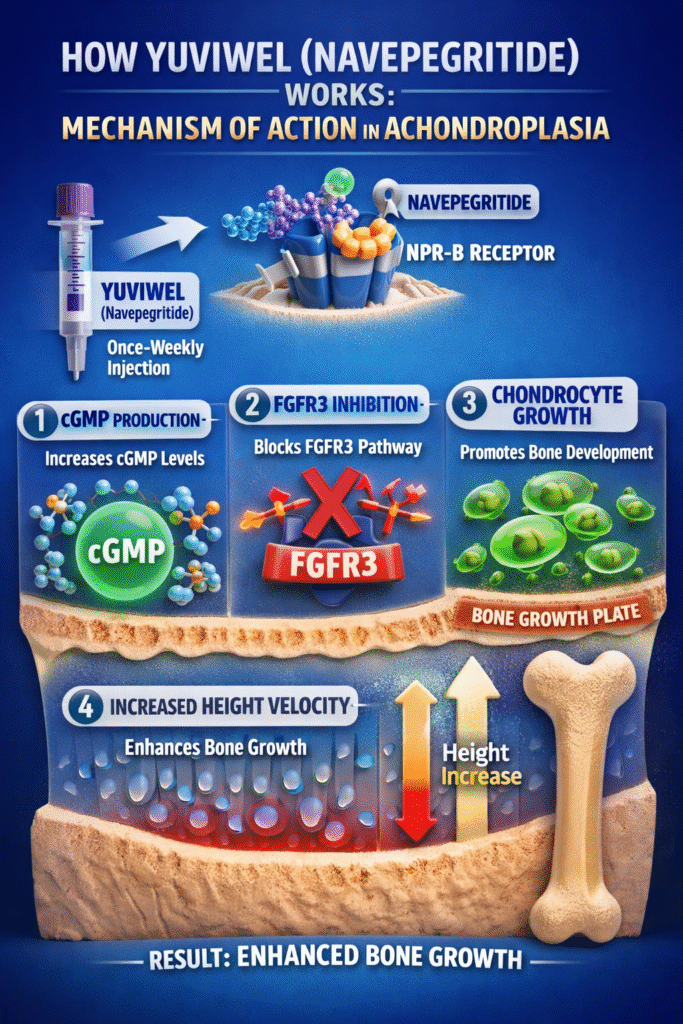
Mechanism of Action (MOA)
The mechanism behind the navepegritide FDA approval is both targeted and highly innovative.
Core Mechanism
Navepegritide acts as a CNP analog, binding to natriuretic peptide receptor-B (NPR-B) located in growth plate cartilage.
Step-by-Step MOA
- Receptor Binding
Navepegritide binds to NPR-B receptors in chondrocytes. - cGMP Production
Activation of NPR-B increases intracellular cyclic guanosine monophosphate (cGMP). - Inhibition of FGFR3 Pathway
Counteracts the overactive FGFR3 signaling seen in achondroplasia. - Promotion of Bone Growth
Enhances chondrocyte proliferation and differentiation. - Increased Height Velocity
Leads to improved longitudinal bone growth.
The navepegritide FDA approval is strongly supported by this precise biological targeting, making it a disease-modifying therapy.
Clinical Efficacy and Trial Data
The navepegritide FDA approval was based on robust clinical trials evaluating its impact on growth velocity.
Key Findings
- Significant increase in annualized height velocity
- Improved skeletal growth patterns
- Sustained efficacy with weekly dosing
Accelerated Approval Basis
- Surrogate endpoint: Height velocity improvement
- Ongoing confirmatory trials required
The navepegritide FDA approval reflects strong clinical evidence supporting its use in pediatric patients.
Pharmacokinetics and Pharmacodynamics
Pharmacokinetics
- Absorption: Slow release from subcutaneous tissue
- Half-life: Extended for weekly dosing
- Distribution: Targets growth plate cartilage
- Metabolism: Peptide degradation
Pharmacodynamics
- Sustained NPR-B activation
- Continuous cGMP signaling
- Long-lasting biological effects
The navepegritide FDA approval ensures consistent therapeutic outcomes with minimal dosing frequency.
Dosage and Administration
The navepegritide FDA approval recommends:
- Route: Subcutaneous injection
- Frequency: Once weekly
- Population: Children ≥2 years
Benefits of Weekly Dosing
- Improved adherence
- Reduced injection burden
- Better quality of life
Side Effects of Navepegritide
The navepegritide FDA approval includes a well-defined safety profile.
Common Side Effects
- Injection site reactions
- Headache
- Vomiting
- Hypotension (due to vasodilatory effect of CNP)
Moderate Side Effects
- Dizziness
- Fatigue
- Gastrointestinal discomfort
Serious Side Effects
- Severe hypotension
- Allergic reactions
The navepegritide FDA approval confirms that most adverse effects are manageable with monitoring.
Drug Interactions
The navepegritide FDA approval highlights potential interactions:
Possible Interactions
- Antihypertensive drugs (enhanced hypotensive effect)
- Diuretics
- Other vasoactive agents
Clinical Recommendations
- Monitor blood pressure
- Adjust concomitant medications if needed
Safety and Precautions
The navepegritide FDA approval includes specific warnings:
Precautions
- Monitor blood pressure regularly
- Use cautiously in cardiovascular conditions
- Ensure open growth plates before initiation
Contraindications
- Hypersensitivity to the drug
- Closed epiphyses
Advantages Over Existing Therapies
The navepegritide offers multiple advantages:
- First once-weekly CNP analog
- Improved compliance vs daily injections
- Targeted molecular therapy
- Better growth outcomes
Impact on Pediatric Endocrinology
The navepegritide FDA approval is a transformative step:
- Provides disease-modifying treatment
- Reduces treatment burden
- Improves long-term outcomes
Future Scope and Ongoing Research
The navepegritide FDA approval opens new research avenues:
- Expanded pediatric indications
- Combination therapies
- Long-term safety studies
Conclusion
The navepegritide represents a groundbreaking advancement in treating achondroplasia. With the approval of Yuviwel, clinicians now have access to a once-weekly, targeted therapy that directly addresses the underlying cause of impaired bone growth.
By improving height velocity and reducing treatment burden, the navepegritide FDA approval sets a new standard in pediatric care. As further studies continue, this therapy has the potential to redefine the management of growth disorders worldwide.

