Table of Contents
Multiple myeloma (MM) is one of the most challenging blood cancers to treat—especially in patients who relapse after multiple treatments. In recent years, treatment options such as proteasome inhibitors, IMIDs, monoclonal antibodies, and CAR-T therapy have extended the lives of patients. However, the disease almost always returns.
This is where Melflufen (melphalan flufenamide) plays an important and innovative role. It is a first-in-class peptide-drug conjugate (PDC) specifically designed to improve drug delivery to cancer cells—especially those that are resistant to standard chemotherapy.
In this comprehensive guide, we explore how Melflufen is an exciting advance in oncology practice for 2025.
What is Melflufen?
Melflufen is a lipophilic alkylating prodrug derived from melphalan. The key innovation is that it utilizes aminopeptidase-driven activation – thereby providing selective cytotoxic activity against malignant plasma cells.
Key features:
• Belongs to a new class of anticancer molecules
• Designed to enhance intracellular delivery of melphalan
• Developed by Oncopeptides AB
• Used in combination with dexamethasone
• Primarily for relapsed or refractory multiple myeloma (RRMM)
Why Melphalan Flufenamide Was Needed in Myeloma Therapy
Advanced multiple myeloma cases often become resistant to standard drugs, including:
| Class of Drug | Examples | Resistance Issue |
| Proteasome Inhibitors | Bortezomib, Carfilzomib | Tumor mutations reduce drug binding |
| IMiDs | Lenalidomide, Pomalidomide | Pathway bypass reduces efficacy |
| Monoclonal Antibodies | Daratumumab, Isatuximab | Loss of antigen expression |
| Traditional Alkylators | Melphalan, Cyclophosphamide | Poor cell penetration |
Melflufen was specifically engineered to overcome these barriers and maintain effectiveness even after multiple lines of prior therapy.
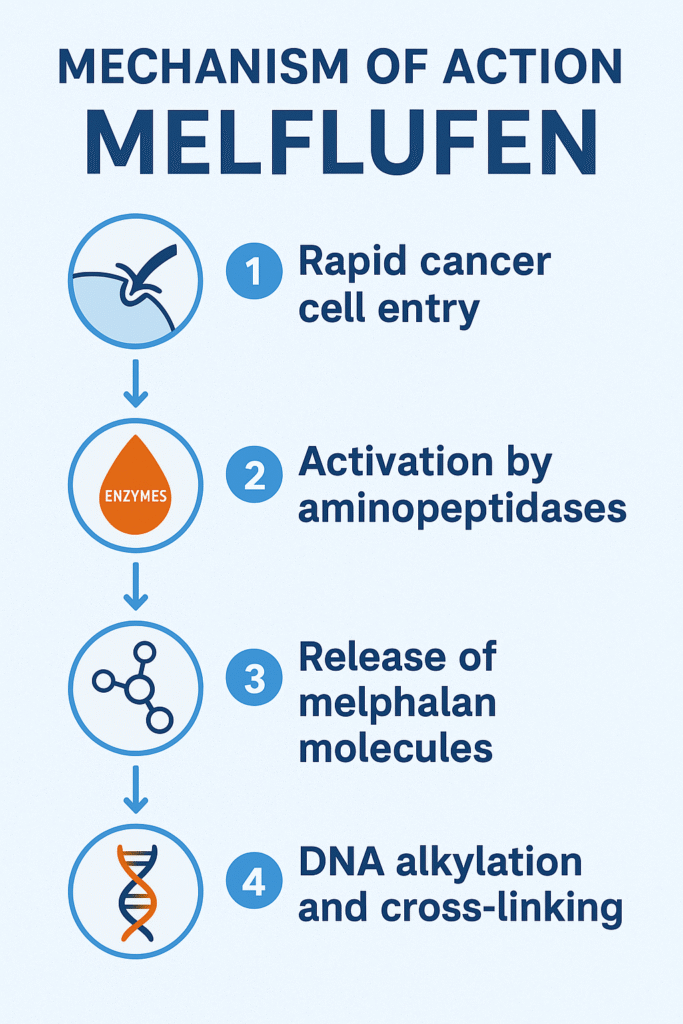
Mechanism of Action — Smart Targeting Technology
Melphalan flufenamide transforms the classical chemotherapy approach into a target-activated weapon. Here’s how:
✓ Step-by-step MOA
1.High penetration into cancer cells
• Melflufen is highly lipophilic → quickly enters tumor cells
2.Cancer cells overexpress aminopeptidases (ENPEP/APN)
3.These enzymes cleave Melflufen → release active Melflufen molecules
4. DNA alkylation and cross-linking
• Active metabolites bind to DNA
• Trigger double-strand breaks
• Activate apoptosis and cell-cycle arrest
✔ Selective killing
✔ More potent than standard Melflufen
✔ Works even in resistant cancer cells
📌 Melflufen is 50 times more cytotoxic in vitro than Melflufen – due to increased intracellular accumulation.
Clinical Evidence
Melphalan Flufenamide has undergone multiple global clinical studies:
O-12-M1 Trial
- Early clinical proof-of-concept study
- Showed successful disease response in heavily treated patients
HORIZON Study
- Included patients with aggressive extramedullary disease
- Demonstrated meaningful responses
- Higher benefit in patients resistant to multiple drug classes
ANCHOR Study
- Evaluated combinations with:
- Daratumumab
- Bortezomib
- Showed synergistic anti-tumor activity
Treatment Outcomes Highlight:
| Outcome | Observation |
| Overall Response Rate (ORR) | Promising in multiple-relapsed cases |
| Duration of Response | Positive in responders |
| Disease Stabilization | Clinically meaningful |
While survival outcomes continue to be studied, these results reinforce Melflufen’s importance for late-stage patients.
Indications:
Melflufen is used in adult MM patients:
✔ After at least 2 prior lines of therapy
✔ Must have been exposed to:
- Anti-CD38 monoclonal antibody
- Proteasome inhibitor (PI)
- IMiD
Ideal for:
- Triple-class refractory MM
- High-risk genetic patients
- Extramedullary progression cases
Administeration
✔ Route: Intravenous infusion
✔ Day 1 of each 28-day cycle
✔ Combined with dexamethasone
Supportive care is typically required to manage blood counts and infection risk.
Benefits
| Advantage | Significance |
| Enhanced internal cell delivery | Faster cancer cell eradication |
| Enzyme-specific activation | Higher tumor selectivity |
| Effective in resistant MM | Fills critical treatment gap |
| Option for patients with no alternatives | Increased survival chances |
Side Effects
Even targeted therapies can involve risks.
Hematological
- Thrombocytopenia
- Neutropenia
- Anemia
🩸 → Frequent blood tests required
Non-hematologic
- Fatigue or weakness
- Pneumonia or respiratory infection
- Peripheral edema
- Gastrointestinal upset (nausea/diarrhea)
- Headache
- Skin reactions
⚠ Monitoring organ function and infection control is essential.
Regulatory & Market Status (2025 Review)
Melphalan Flufenamide initially received accelerated approval in some regions due to its benefits in late-stage MM.
However, regulatory review is ongoing globally to ensure:
🔹 Stronger survival impact
🔹 Balanced benefit-risk ratio
Despite evaluation changes, many clinicians still prefer Melphalan Flufenamide in selective, aggressive disease situations where no alternatives exist.
Melflufen vs Melphalan: Key Differences
| Parameter | Melflufen | Melphalan |
| Cell Penetration | Much higher | Limited |
| Selectivity | Tumor-targeted | Broad, non-specific |
| Activation | Enzyme-dependent ↑ | Systemic activation |
| Potency | Higher cancer killing | Standard effect |
| Resistance Handling | Better | Limited in resistant disease |
📌 That’s whyMelphalan Flufenamide is often referred to as precision melphalan.
Role of Melphalan Flufenamide in Future Oncology
Melflufen represents a platform technology, offering future potential in:
🔬 Solid tumors with high aminopeptidase expression
🧬 Combination with next-gen immunotherapies
⏳ Earlier line therapy if survival outcomes improve
🎯 Personalized cancer therapy strategies
Researchers are currently exploring its utility in:
- High-risk cytogenetics
- Extramedullary relapse cases
- Combinations with CAR-T sequencing
Best Practices and Precautions (Educational Only)
For safe therapeutic outcomes:
✔ Routine CBC monitoring
✔ Dose adjustment in renal impairment
✔ Infection prevention strategy
✔ Manage thrombocytopenia early
✔ Personalized regimen based on tolerance
⚠ Patients must follow oncologist-guided treatment decisions only.
Melflufen for Pharmacy Students: Key Learning Concepts
📌 Drug transport innovations
📌 Enzyme-activated prodrugs
📌 Managing hematologic toxicity
📌 Combination therapy in RRMM
📌 Translational research into clinical oncology
Understanding Melphalan Flufenamide helps future pharmacists appreciate how drug design enhances precision cancer medicine.
Conclusion: Why Melflufen Remains an Important Option in 2025
Melflufen has changed the face of drug-resistant multiple myeloma:
✔ Delivering chemotherapy more efficiently to cancer cells
✔ Providing benefit when conventional therapies fail
✔ Showing activity against aggressive extramedullary tumors
While regulatory scrutiny is ongoing, real-world experience demonstrates its value as a life-prolonging therapy in advanced MM.
Melflufen’s journey reflects the spirit of innovation – transforming a classic drug into a modern precision therapy that offers new hope to patients at the most difficult stages of their cancer battle.

