The landscape of medicine is continually being reshaped by groundbreaking scientific advancements. Few areas have seen a more profound transformation than the treatment of rare genetic disorders. Among these innovations, the lumasiran mechanism of action stands out as a paradigm shift in the management of Primary Hyperoxaluria Type 1 (PH1), a devastating, ultra-rare disease.Lumasiran, marketed as OXLUMO, is not a traditional small-molecule drug; it is a sophisticated RNA interference (RNAi) therapeutic that targets the very root of the disease’s pathology. This comprehensive analysis will explore the intricate science behind this drug, detailing its mechanism, its clinical validation, and its profound impact on patients’ lives.
Understanding Primary Hyperoxaluria Type 1 (PH1): The Need for Lumasiran Mechanism of Action
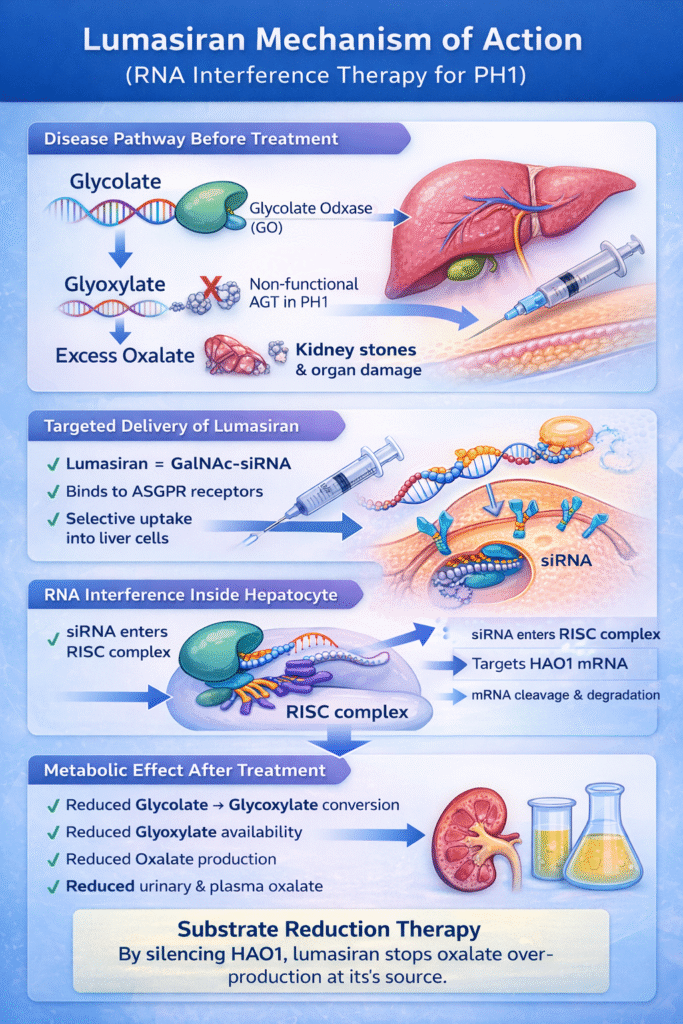
To fully appreciate the elegance of the lumasiran mechanism of action, one must first understand the metabolic catastrophe that is Primary Hyperoxaluria Type 1 (PH1). PH1 is an autosomal recessive disorder caused by a genetic defect in the AGXT gene [1]. This gene provides instructions for making the enzyme alanine:glyoxylate aminotransferase (AGT), which is primarily active in the liver.
The Metabolic Defect
The AGT enzyme is crucial for detoxifying a compound called glyoxylate. Normally, AGT converts glyoxylate into the harmless amino acid glycine. In patients with PH1, the AGT enzyme is either deficient or non-functional. This metabolic block causes glyoxylate to accumulate.
The Pathological Consequence: Oxalate Overproduction
The accumulated glyoxylate is then shunted down an alternative metabolic pathway, where it is converted into oxalate. Oxalate is a waste product that the body cannot metabolize and must excrete via the kidneys. While healthy individuals produce small amounts of oxalate, PH1 patients suffer from a massive, chronic overproduction of oxalate in the liver.
This excessive oxalate is filtered by the kidneys, where it combines with calcium to form highly insoluble calcium oxalate crystals. This leads to a cascade of severe complications:
- Nephrocalcinosis: The deposition of calcium oxalate crystals within the kidney tissue.
- Nephrolithiasis: The formation of painful kidney stones.
- End-Stage Renal Disease (ESRD): Over time, the relentless crystal deposition destroys kidney function, leading to kidney failure.
- Systemic Oxalosis: Once the kidneys fail, oxalate can no longer be excreted and begins to deposit in other organs, including the eyes, bones, heart, and blood vessels, leading to multi-organ dysfunction and often, premature death.
Prior to the advent of RNAi therapeutics, treatment for PH1 was limited to hyperhydration and high-dose pyridoxine (Vitamin B6) for a small subset of responders. Ultimately, a combined liver and kidney transplant was necessary, a major surgical procedure with significant risks and lifelong immunosuppression. The need for a targeted, non-surgical intervention was immense.
The Scientific Foundation: How RNA Interference Powers the Lumasiran Mechanism of Action
Lumasiran belongs to a revolutionary class of drugs known as small interfering RNA (siRNA) therapeutics, which harness the natural biological process of RNA interference (RNAi). RNAi is a fundamental cellular mechanism that regulates gene expression by silencing specific messenger RNA (mRNA) molecules.
How siRNA Works in Lumasiran Mechanism of action
The central dogma of molecular biology states that DNA is transcribed into mRNA, which is then translated into protein. siRNA molecules intercept this process.
- Introduction: An siRNA therapeutic, like lumasiran, is introduced into the cell.
- RISC Complex: The siRNA is incorporated into a cellular machinery called the RNA-induced Silencing Complex (RISC).
- Target Recognition: The RISC complex uses one strand of the siRNA (the guide strand) to precisely locate a complementary target mRNA sequence.
- Cleavage: Once the target mRNA is found, the RISC complex cleaves and destroys it.
- Silencing: With the mRNA destroyed, the cell can no longer produce the corresponding protein. The gene is effectively “silenced.”
This Lumasiran Mechanism of action offers unprecedented specificity, allowing scientists to turn off the production of a single, disease-causing protein without affecting other cellular processes.
The Precision of Lumasiran Mechanism of Action: Targeting the Liver
The brilliance of the lumasiran mechanism of action lies in its strategic targeting of an enzyme upstream of the primary genetic defect. Instead of trying to fix the non-functional AGT enzyme, lumasiran focuses on reducing the substrate that leads to oxalate overproduction.
Targeting the HAO1 Gene
lumasiran mechanism of action is specifically designed to target the messenger RNA (mRNA) of the Hydroxyacid Oxidase 1 (HAO1) gene [2]. The HAO1 gene provides the instructions for making the enzyme Glycolate Oxidase (GO).
The metabolic pathway leading to oxalate is as follows:
The metabolic pathway of oxalate production in the liver of a patient with Primary Hyperoxaluria Type 1 (PH1). It illustrates how the disease causes a toxic buildup of oxalate through a series of enzymatic steps
| Component | Description |
| Glycolate | The starting substrate in this specific metabolic chain. |
| Glycolate Oxidase (GO) | The enzyme that converts Glycolate into Glyoxylate. This is the specific target that Lumasiran inhibits. |
| Glyoxylate | An intermediate compound. In healthy individuals, it is detoxified by the AGT enzyme. |
| Non-functional AGT | The core genetic defect in PH1. Because the AGT enzyme doesn’t work, it cannot convert Glyoxylate into the harmless amino acid glycine. |
| Oxalate | The final toxic byproduct. Because the “detox” route is blocked by the non-functional AGT, the body is forced to turn Glyoxylate into Oxalate, leading to kidney stones and organ damage. |
How Lumasiran Intervenes
lumasiran mechanism of action works by “silencing” the production of the Glycolate Oxidase (GO) enzyme. By removing the GO enzyme from the equation, the body cannot convert Glycolate into Glyoxylate. Without Glyoxylate, the “Non-functional AGT” has nothing to turn into Oxalate, effectively stopping the overproduction at the source.
In PH1 patients, the non-functional AGT allows glyoxylate to be converted into oxalate. Lumasiran intervenes at the very first step of this sequence.
The lumasiran mechanism of action can be broken down into three critical steps:
Delivery, silencing, and Metabolic effect.
The lumasiran mechanism of action can be broken down into three critical steps: delivery, silencing, and metabolic effect.
1. Targeted Delivery via GalNAc Conjugation
A major challenge for any RNAi therapeutic is delivering the fragile siRNA molecule to the correct cell type. Lumasiran overcomes this through a proprietary delivery system involving N-acetylgalactosamine (GalNAc) conjugation [3].
- The Liver as the Factory: Since the liver is the sole site of oxalate overproduction, it is the only organ that needs to be targeted.
- The GalNAc Key: Lumasiran is chemically linked to three GalNAc sugar molecules.
- The ASGPR Lock: Hepatocytes (liver cells) express a receptor called the asialoglycoprotein receptor (ASGPR) on their surface. This receptor has a high affinity for GalNAc.
- Internalization: When Lumasiran is injected subcutaneously, the GalNAc conjugate binds to the ASGPR, and the entire complex is internalized into the hepatocyte via endocytosis.
This highly specific delivery system ensures that the drug is concentrated in the liver, maximizing its therapeutic effect while minimizing off-target effects in other tissues.
2. Gene Silencing of HAO1
Once inside the hepatocyte, the siRNA component of lumasiran is released and loaded into the RISC complex. The guide strand then seeks out and destroys the HAO1 mRNA.
- The Result: The destruction of HAO1 mRNA prevents the production of the Glycolate Oxidase (GO) enzyme.
- The Effect: The GO enzyme level in the liver is significantly reduced, leading to a profound decrease in its activity.
3. The Metabolic Cascade: Substrate Reduction
With the GO enzyme inhibited, the conversion of glycolate to glyoxylate is drastically reduced. This is the ultimate goal of the lumasiran mechanism of action.
- Reduced Glyoxylate: Less glyoxylate is produced.
- Reduced Oxalate: With less glyoxylate available as a substrate, the non-functional AGT pathway has less material to convert into oxalate.
- The Outcome: The hepatic overproduction of oxalate is dramatically lowered, leading to a sustained reduction in both urinary oxalate (UOx) and plasma oxalate (POx) levels.
This lumasiran mechanism of action strategy is known as substrate reduction therapy. By cutting off the supply chain of the toxic metabolite (oxalate), lumasiran effectively bypasses the genetic defect, offering a functional cure for the metabolic imbalance.
Pharmacokinetics and Pharmacodynamics: The Clinical Impact of Lumasiran Mechanism of Action
The clinical success of the lumasiran mechanism of action is rooted in its favorable pharmacokinetics (PK) and pharmacodynamics (PD).
Pharmacokinetics (PK)
Lumasiran Mechanism of action is administered via a simple subcutaneous injection. The GalNAc conjugation allows for rapid uptake by the liver.
- Absorption: Rapidly absorbed after subcutaneous injection.
- Distribution: Highly concentrated in the liver due to the ASGPR targeting.
- Metabolism and Excretion: The siRNA is metabolized by nucleases, and the resulting components are primarily excreted in the urine.
The PK profile allows for a convenient dosing schedule, typically involving an initial loading phase followed by monthly maintenance doses, which significantly improves patient compliance compared to daily oral medications.
Pharmacodynamics (PD)
The pharmacodynamic effect is the sustained reduction in oxalate levels, which is directly proportional to the degree of HAO1 gene silencing.
- Rapid Onset: Clinical trials demonstrated a rapid and significant reduction in UOx levels within the first month of treatment.
- Sustained Effect: The RNAi mechanism provides a durable effect. Once the HAO1 mRNA is silenced, it takes time for new mRNA to be transcribed and for GO enzyme levels to recover. This allows for the monthly or quarterly dosing schedule, maintaining a consistently low level of oxalate production.
- Clinical Endpoint: The primary PD endpoint in clinical trials was the percentage change from baseline in 24-hour UOx excretion, which showed a median reduction of over 50% in most patient populations [4].
Clinical Validation: The ILLUMINATE Program
The efficacy and safety of lumasiran were rigorously established through the ILLUMINATE clinical trial program, which included a diverse range of PH1 patients followed by the lumasiran mechanism of action.
ILLUMINATE-A: The Pivotal Trial
The Phase 3 ILLUMINATE-A trial was the pivotal study that led to regulatory approval. It was a randomized, double-blind, placebo-controlled trial in patients aged six years and older with PH1 and relatively preserved kidney function.
- Key Finding: Patients treated with lumasiran achieved a statistically significant and clinically meaningful reduction in 24-hour UOx excretion compared to the placebo group. The majority of patients achieved near-normal or normal UOx levels.
- Clinical Benefit: This reduction in oxalate is expected to slow or halt the progression of kidney damage and reduce the incidence of kidney stone events.
ILLUMINATE-B: Pediatric Efficacy
Recognizing the aggressive nature of PH1 in infants and young children, the ILLUMINATE-B trial focused on patients younger than six years.
- Key Finding: Lumasiran demonstrated similar efficacy in this younger population, with rapid and sustained reductions in the spot UOx:creatinine ratio, a common measure in pediatric patients.
- Significance: Early intervention is critical in PH1, and the success of lumasiran in this group offers a chance to prevent the most severe, life-threatening complications of the disease.
ILLUMINATE-C: Advanced Disease
The ILLUMINATE-C trial addressed the most challenging patient population: those with advanced kidney disease, including patients on dialysis. In these patients, the kidneys cannot effectively excrete oxalate, leading to high plasma oxalate (POx) levels and systemic oxalosis.
- Key Finding: Lumasiran treatment led to a sustained reduction in POx levels.
- Impact: Reducing the systemic oxalate burden is crucial for preventing or reversing the deposition of calcium oxalate in non-renal tissues, a major cause of morbidity and mortality in advanced PH1.
Across all trials, lumasiran was generally well-tolerated. The most common adverse events were mild and transient injection site reactions, consistent with the highly targeted nature of the drug.
The introduction of the lumasiran mechanism of action has fundamentally changed the treatment algorithm for PH1.
The introduction of Lumasiran Mechanism of action has fundamentally changed the treatment algorithm for PH1.
Comparison with Traditional Therapies
| Therapy | Mechanism of Action | Efficacy in PH1 | Limitations |
| Hyperhydration | Increases urine volume to dilute oxalate | Supportive, does not address overproduction | Difficult compliance, risk of fluid overload |
| Pyridoxine (Vitamin B6) | Acts as a cofactor for AGT, improving function in some patients | Effective in only ~30% of PH1 patients | Only works for specific AGXT mutations |
| Combined Liver/Kidney Transplant | Replaces the defective AGT enzyme (liver) and damaged organ (kidney) | Curative, but high risk and resource-intensive | Major surgery, lifelong immunosuppression, organ scarcity |
| Lumasiran (OXLUMO) | RNAi-mediated HAO1 gene silencing | High efficacy in all PH1 patients, regardless of mutation | Requires ongoing injection, long-term data still accumulating |
Comparison with Other RNAi Agents
Lumasiran Mechanism of action is not the only RNAi therapeutic developed for hyperoxaluria. Another agent, nedosiran, is also in development. While both use the RNAi platform and target the liver to reduce oxalate production, their specific targets differ.
- Lumasiran: Targets HAO1 mRNA (Glycolate Oxidase).
- Nedosiran: Targets LDHA mRNA (Lactate Dehydrogenase A).
Both strategies aim to reduce the amount of glyoxylate available for conversion to oxalate, but the subtle differences in their lumasiran mechanism of action and other agents may offer clinicians options based on patient-specific factors.
The Future of Substrate Reduction Therapy
The success of the lumasiran mechanism of action is a testament to the power of precision medicine. It demonstrates that targeting an upstream enzyme in a metabolic pathway can be a highly effective strategy for treating genetic disorders, even when the primary defective enzyme cannot be directly fixed.
The long-term implications of this therapy are profound. By consistently lowering the oxalate burden, lumasiran offers the potential to:
- Prevent the progression of kidney disease in newly diagnosed patients.
- Stabilize or even improve kidney function in patients with moderate disease.
- Reduce the systemic oxalate deposits in patients with advanced disease, potentially delaying or eliminating the need for complex, high-risk organ transplantation.
As research continues, the focus will shift to understanding the full extent of renal recovery and the long-term quality of life improvements afforded by this revolutionary treatment. The lumasiran mechanism of action has not only provided a new drug but has also validated the entire RNAi platform as a powerful tool in the fight against genetic disease.
Conclusion
Lumasiran represents a significant advancement in treating Primary Hyperoxaluria Type 1 through RNA interference and targeted GalNAc delivery, effectively silencing the HAO1 gene to inhibit Glycolate Oxidase. This mechanism reduces glyoxylate production, leading to substantial decreases in toxic oxalate levels across all ages and kidney function stages, establishing a new standard of care and hope for patients and families.

