Table of Contents
Introduction: A New Chapter in Cancer Immunotherapy
Cancer treatment has changed dramatically over the past few decades, moving beyond chemotherapy and radiation to therapies that harness the body’s own immune system. Among these next-generation approaches, Lifileucel immunotherapy is being hailed as a personalized, cell-based treatment that offers hope to patients with advanced cancer – especially those who have exhausted conventional options.
Approved by the US FDA in 2024 under the brand name Amtagvi™, Lifileucel represents a pioneering tumor-infiltrating lymphocyte (TIL) therapy designed to revive the immune system’s natural ability to recognize and destroy tumor cells. This advancement opens up new possibilities for treating metastatic melanoma and potentially other difficult-to-treat cancers.
What Is Lifileucel Immunotherapy?
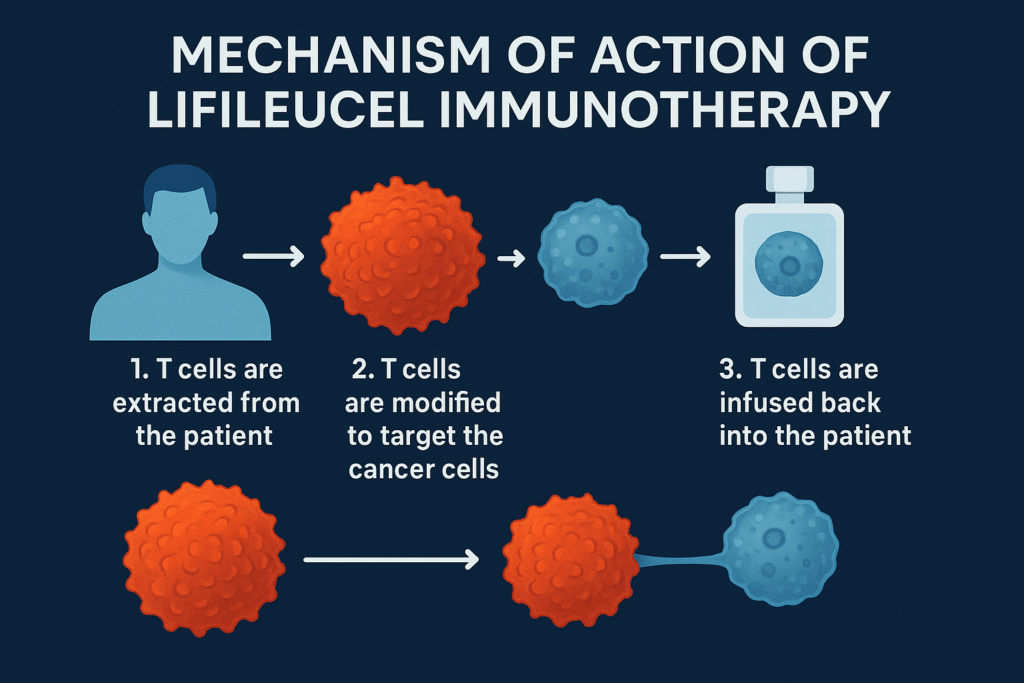
Lifileucel immunotherapy is a type of adoptive cell therapy (ACT) that uses a patient’s own immune cells – specifically tumor-infiltrating lymphocytes (TILs) – to fight cancer. TILs are naturally occurring immune cells that enter a patient’s tumor, indicating that the body has already tried to mount an immune response against the cancer. However, these cells are often overwhelmed by the tumor’s suppressive environment and lose their effectiveness.
Lifileucel works by:
1. Extracting TILs from a sample of the patient’s tumor tissue.
2. Expanding these cells in large numbers in the laboratory using special growth factors.
3. Re-infusing the expanded TILs into the patient after a conditioning chemotherapy regimen.
Once injected, these reactivated immune cells seek out and attack cancer cells throughout the body, using their power to counteract the patient’s own immune system.
The Science Behind Lifileucel: Understanding TIL Therapy
At its core, lifileucel immunotherapy is built on the science of immune reactivation. Here’s how it works step-by-step:
1. Tumor Resection and Cell Harvesting
A small piece of tumor tissue is surgically removed. This sample contains both cancer cells and tumor-infiltrating lymphocytes—immune cells that have already recognized tumor antigens.
2. Ex Vivo Expansion
In a controlled laboratory setting, scientists separate the TILs and culture them with interleukin-2 (IL-2), a cytokine that stimulates immune cell growth. Over two to three weeks, billions of activated TILs are produced.
3. Patient Conditioning
Before reintroducing these TILs, patients undergo lymphodepleting chemotherapy to remove suppressive immune cells and make room for the infused TILs to thrive.
4. TIL Infusion
The expanded TILs are then infused back into the patient intravenously. These cells recognize cancer-specific antigens and aggressively attack tumor cells.
5. IL-2 Support
After infusion, patients receive low doses of IL-2 to support TIL survival and activation in the body.
This personalized approach allows for targeted immune activity that adapts to each patient’s unique tumor profile, setting lifileucel apart from conventional immunotherapies.
Mechanism of Action: How Lifileucel Targets Cancer
The mechanism of action of lifileucel revolves around leveraging naturally occurring T-cell responses. Here’s a simplified breakdown:
- Tumor Recognition: The harvested TILs contain T cells that have already identified cancer antigens.
- Activation and Expansion: Lab processing amplifies these cells’ numbers and potency.
- Reinfusion and Attack: When infused back into the body, these activated T cells locate tumor sites and destroy cancer cells by releasing cytotoxic molecules like perforin and granzymes.
- Immune Memory: Some of these cells persist as memory T cells, offering ongoing surveillance against cancer recurrence.
This mechanism differs from checkpoint inhibitors (like pembrolizumab or nivolumab), which remove immune “brakes”, whereas lifileucel directly boosts the number and activity of tumor-targeting cells.
Clinical Applications of Lifileucel Immunotherapy
1. FDA-Approved Indication: Advanced Melanoma
As of 2024, the FDA approved lifileucel (Amtagvi™) for the treatment of unresectable or metastatic melanoma in adults who have previously received PD-1 inhibitors and, if applicable, BRAF/MEK inhibitors.
Clinical trials such as C-144-01 demonstrated durable responses in patients who had failed multiple lines of therapy, making lifileucel a vital new option for those with limited alternatives.
2. Ongoing Trials in Other Cancers
Beyond melanoma, clinical research is exploring lifileucel’s potential in:
- Cervical cancer
- Head and neck squamous cell carcinoma (HNSCC)
- Non-small cell lung cancer (NSCLC)
- Breast cancer
- Ovarian cancer
These studies aim to determine whether TIL therapy’s success in melanoma can be extended to other solid tumors, which have historically been challenging to treat with immunotherapy.
Patient Eligibility and Treatment Process
Not every patient qualifies for lifileucel immunotherapy. Eligibility depends on:
- Having advanced or metastatic melanoma unresponsive to standard therapies
- Adequate organ function and performance status to undergo surgery and chemotherapy
- Absence of rapidly progressing disease that would preclude the time needed for cell preparation
Treatment Timeline Overview
- Tumor Resection (Day 0–2): A small tumor sample is collected.
- TIL Manufacturing (Weeks 2–3): The sample is sent to a manufacturing facility for expansion.
- Lymphodepletion (Few Days): The patient receives chemotherapy to prepare the immune environment.
- TIL Infusion (Day 0): Lifileucel is infused intravenously.
- IL-2 Support (Few Days): Post-infusion support to enhance cell persistence.
The overall treatment process typically spans 4–6 weeks from tumor collection to infusion.
Benefits of Lifileucel Immunotherapy
1. Personalized and Targeted
Unlike generalized chemotherapy, lifileucel immunotherapy is patient-specific, targeting each individual’s unique tumor antigens.
2. Effective in Resistant Tumors
Lifileucel has shown efficacy in patients who did not respond to checkpoint inhibitors or targeted therapies, offering hope to those with refractory melanoma.
3. Durable Responses
Clinical data reveal long-lasting tumor regressions, with some patients maintaining remission for over two years post-treatment.
4. Immune Memory and Long-Term Surveillance
By reintroducing activated T cells capable of forming immune memory, lifileucel may help prevent recurrence.
5. Potential for Broader Cancer Use
Ongoing studies may extend its benefits to other solid tumors, revolutionizing cancer immunotherapy.
Risks and Side Effects of Lifileucel Immunotherapy
While effective, lifileucel immunotherapy can cause immune-related and chemotherapy-related side effects. Common adverse events include:
- Fatigue and fever
- Nausea, vomiting, or diarrhea
- Low blood counts due to lymphodepleting chemotherapy
- Hypotension (low blood pressure) during or after infusion
- Cytokine release-related symptoms (fever, chills, body aches)
- Infections due to temporary immune suppression
Most side effects are manageable under medical supervision, and patients are usually hospitalized during and after infusion for monitoring.
Recent Research and Clinical Developments
1. Pivotal C-144-01 Trial Results
In this Phase II trial involving patients with advanced melanoma, lifileucel demonstrated an objective response rate (ORR) of ~31%, with some achieving complete remission. Importantly, responses were durable, lasting over 18 months in many cases.
2. Combination Studies
Researchers are exploring combining lifileucel with:
- Checkpoint inhibitors (like pembrolizumab) to enhance efficacy
- Targeted therapies for synergistic tumor killing
- New cytokine formulations to improve TIL persistence
3. Manufacturing Advances
The development of cryopreservation and streamlined manufacturing techniques has reduced production time, making the therapy more accessible and scalable for commercial use.
Challenges and Limitations
Despite its promise, lifileucel immunotherapy faces several hurdles:
- Complex Manufacturing: Producing patient-specific TILs is labor-intensive and time-consuming.
- High Cost: Personalized cell therapies remain expensive due to manufacturing and hospitalization costs.
- Limited Access: Currently, treatment centers offering TIL therapy are few.
- Variable Response Rates: Not all patients respond equally, and biomarkers for predicting outcomes are still under study.
Researchers and biotech companies are working to optimize scalability, reduce cost, and identify predictive biomarkers to enhance treatment success.
Future Perspectives: The Road Ahead for Lifileucel
The future of lifileucel immunotherapy looks highly promising. With expanded clinical trials across multiple cancers, manufacturing innovations, and potential combinatorial regimens, TIL therapy could become a mainstay of solid tumor treatment in the next decade.
Emerging trends include:
- Allogeneic TIL platforms (off-the-shelf versions)
- AI-driven cell selection to improve efficacy prediction
- Integration with mRNA technology for immune enhancement
As the field matures, lifileucel could lead the way toward personalized, durable, and potentially curative cancer immunotherapy.
Conclusion: A New Hope in Cancer Treatment
Lifileucel immunotherapy represents a revolutionary advance in oncology – combining the precision of cellular medicine with the power of the immune system. By reawakening a patient’s own tumor-fighting cells, Lifileucel has redefined what is possible for individuals battling advanced melanoma and may soon extend its benefits to other cancers.
Despite challenges in accessibility, cost, and standardization, the clinical success and ongoing innovation around Lifileucel immunotherapy signal a new era of personalized cancer care – where hope is built and healing begins at the cellular level.
Key Takeaways
- Lifileucel immunotherapy uses a patient’s own tumor-infiltrating lymphocytes (TILs) to fight cancer.
- FDA-approved in 2024 for advanced melanoma (brand name: Amtagvi™).
- Offers durable responses in treatment-resistant cancers.
- Associated with manageable side effects under hospital supervision.
- Future developments may expand its use to lung, cervical, and ovarian cancers.


1 thought on “Lifileucel Immunotherapy: A Breakthrough in Personalized Cancer Treatment (2025 Guide)”