Table of Contents
Can a disease rooted in toxic metabolite accumulation be treated by silencing a single gene?
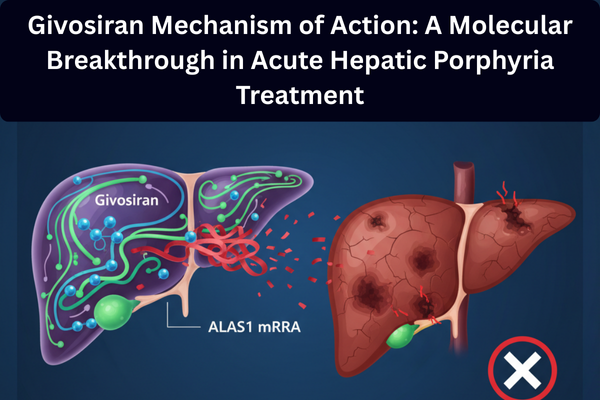
The answer is yes. The givosiran mechanism of action represents a paradigm shift in treating acute hepatic porphyria by targeting the disease at its molecular origin. Unlike traditional symptomatic treatments, givosiran works through RNA interference to suppress the overproduction of toxic heme intermediates. For pharmacists, clinicians, and patients alike, understanding how givosiran works is key to appreciating its role in modern precision medicine.
Overview of Acute Hepatic Porphyria (AHP)
Acute hepatic porphyria is a group of rare, inherited metabolic disorders characterized by defects in hepatic heme biosynthesis. These defects lead to the accumulation of neurotoxic intermediates, primarily delta-aminolevulinic acid (ALA) and porphobilinogen (PBG).
Patients with AHP experience:
- Severe abdominal pain
- Peripheral neuropathy
- Autonomic dysfunction
- Psychiatric symptoms
- Life-threatening acute attacks
AHP attacks are unpredictable and recurrent, often triggered by stress, medications, fasting, or hormonal changes. Traditional therapies focus on treating acute attacks but fail to prevent disease progression. This is where the givosiran mechanism of action offers a disease-modifying solution.
What Is Givosiran?
Givosiran is a small interfering RNA (siRNA) therapeutic designed specifically to treat acute hepatic porphyria. It belongs to a novel class of RNA interference medicines that selectively silence disease-causing genes in the liver.
Rather than replacing enzymes or neutralizing toxins, givosiran directly suppresses the overactive enzyme responsible for toxic metabolite production. This targeted approach makes the givosiran mechanism of action unique among porphyria treatments.
FDA Approval Status and Regulatory Background
Givosiran received regulatory approval for the treatment of adults with acute hepatic porphyria based on its ability to significantly reduce the frequency of acute attacks. Its approval marked one of the first RNA interference therapies for a rare metabolic disorder.
The regulatory decision recognized givosiran as:
- A first-in-class RNAi therapy for AHP
- A disease-modifying treatment rather than symptomatic care
- A major advancement in rare disease pharmacotherapy
Post-approval monitoring continues to support its long-term effectiveness and safety.
Detailed Givosiran Mechanism of Action (Step-by-Step)
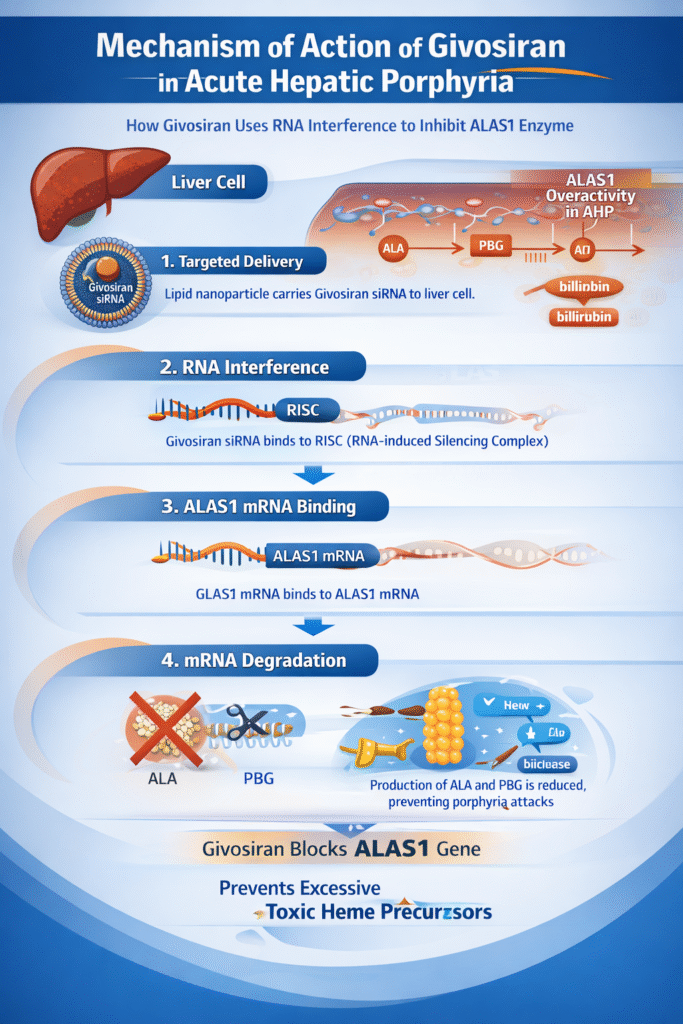
The givosiran mechanism of action is based on RNA interference, a natural cellular process that regulates gene expression.
Step 1: Targeted Liver Delivery
Givosiran is administered subcutaneously and selectively taken up by hepatocytes, the primary site of heme synthesis. Its molecular design ensures liver-specific action.
Step 2: RNA Interference Activation
Once inside the liver cell, givosiran releases siRNA molecules that bind to the RNA-induced silencing complex (RISC).
Step 3: ALAS1 mRNA Binding
The siRNA specifically recognizes and binds to messenger RNA encoding aminolevulinic acid synthase 1 (ALAS1), the rate-limiting enzyme in hepatic heme production.
Step 4: mRNA Degradation
The bound ALAS1 mRNA is cleaved and degraded, preventing translation into the ALAS1 enzyme.
Step 5: Reduced Toxic Metabolites
With ALAS1 suppressed, production of ALA and PBG drops significantly, preventing neurotoxicity and acute porphyria attacks.
This precise gene-silencing process defines the givosiran mechanism of action and explains its high clinical efficacy.
Role of ALAS1 Inhibition in Porphyria
ALAS1 is the first and rate-limiting enzyme in the heme biosynthesis pathway. In AHP, defective downstream enzymes cause feedback dysregulation, leading to ALAS1 overactivation.
By inhibiting ALAS1:
- Toxic precursors are prevented from forming
- Hepatic metabolic balance is restored
- Neurological damage is reduced
The givosiran mechanism of action directly addresses this biochemical imbalance.
How Givosiran Reduces Toxic Heme Intermediates
Givosiran suppresses excessive heme precursor production rather than attempting to remove them after formation. This upstream intervention results in:
- Sustained reduction in ALA and PBG levels
- Prevention of neuronal injury
- Fewer hospitalizations
- Improved long-term outcomes
This preventive mechanism distinguishes givosiran from traditional porphyria therapies.
Indications and Approved Uses
Givosiran is indicated for:
- Adults diagnosed with acute hepatic porphyria
- Patients with recurrent acute attacks
- Individuals requiring long-term attack prevention
It is not intended for other types of porphyria unrelated to hepatic ALAS1 dysregulation.
Dosage and Administration
- Route: Subcutaneous injection
- Dose: Weight-based monthly dosing
- Administration: Healthcare professional or trained patient
Consistent dosing is essential to maintain suppression of ALAS1 activity and sustain the givosiran mechanism of action.
Pharmacokinetics and Pharmacodynamics
Pharmacokinetics
- Rapid hepatic uptake
- Minimal systemic exposure
- Predictable monthly plasma clearance
Pharmacodynamics
- Sustained ALAS1 suppression
- Long-term reduction in neurotoxic metabolites
- Stable biochemical control with repeated dosing
There is no significant accumulation, supporting chronic use.
Clinical Trial Evidence and Efficacy Outcomes
Clinical evaluation demonstrated:
- Significant reduction in annualized attack rate
- Decreased need for hospitalization
- Reduced opioid use
- Improved patient-reported outcomes
The consistency of these results validates the therapeutic value of the givosiran mechanism of action.
Benefits of Givosiran Therapy in AHP
Key benefits include:
- Disease modification rather than symptomatic relief
- Prevention of acute attacks
- Improved quality of life
- Reduced healthcare burden
For pharmacists, givosiran represents a shift toward molecularly targeted therapy in metabolic disease.
Side Effects and Safety Profile
Common adverse effects:
- Injection site reactions
- Nausea
- Fatigue
Less common but clinically important:
- Elevated liver enzymes
- Renal function changes
Ongoing monitoring ensures safe long-term use while preserving the benefits of the givosiran mechanism of action.
Warnings, Precautions, and Contraindications
- Monitor liver and renal function regularly
- Use caution in patients with pre-existing hepatic impairment
- Avoid use in patients with known hypersensitivity
Pharmacists play a critical role in safety surveillance.
Drug Interactions
Givosiran has minimal drug-drug interaction potential due to:
- Lack of CYP450 metabolism
- Liver-specific RNAi activity
However, hepatotoxic drugs should be used cautiously.
Comparison With Other Porphyria Treatments
| Treatment | Target | Prevention | Limitation |
| Givosiran | ALAS1 gene | Yes | Monitoring needed |
| Hemin | Heme replacement | No | Acute only |
| Supportive care | Symptoms | No | Disease progression |
The givosiran mechanism of action uniquely targets the root cause.
Patient Counseling Points (Pharmacy-Focused)
Pharmacists should counsel patients on:
- Importance of monthly adherence
- Recognizing early attack symptoms
- Avoiding known triggers
- Reporting adverse effects promptly
Education reinforces therapeutic success.
Future Scope and RNAi Research
The success of givosiran has accelerated:
- RNAi drug development
- Expansion into other metabolic diseases
- Personalized gene-silencing therapies
The givosiran mechanism of action serves as a model for next-generation medicines.
Conclusion
So, can acute hepatic porphyria be treated at the molecular level?
Absolutely. The givosiran mechanism of action demonstrates how precise gene silencing can transform a devastating metabolic disorder into a manageable condition. By targeting ALAS1 at its source, givosiran moves beyond symptom control to true disease modification—setting a new benchmark in RNA interference therapy and pharmacy-driven precision medicine.