Table of Contents
The approval of Waskyra (etuvetidigene autotemcel) by the U.S. Food and Drug Administration represents a defining moment in the treatment of rare genetic immunodeficiency disorders. As the first FDA-approved cell-based gene therapy for Wiskott-Aldrich syndrome (WAS), this therapy introduces a new era of personalized, gene-corrective medicine for patients who previously had limited therapeutic options.
Wiskott-Aldrich syndrome is a serious inherited disorder that affects immune function, blood clotting, and inflammatory responses. For decades, treatment strategies focused primarily on managing symptoms or pursuing hematopoietic stem cell transplantation (HSCT) when a suitable donor was available. However, donor availability has long remained a critical limitation. The approval of etuvetidigene autotemcel directly addresses this challenge by offering a gene therapy option for patients without a matched related donor.
This article provides an in-depth, comprehensive overview of etuvetidigene autotemcel, including its clinical role, patient eligibility, treatment process, mechanism of action, safety considerations, regulatory approval, and long-term implications for patients and healthcare systems.
Understanding Wiskott-Aldrich Syndrome (WAS)
Wiskott-Aldrich syndrome is a rare, inherited, X-linked genetic disorder caused by mutations in the WAS gene. This gene plays a crucial role in regulating immune cell signaling, cytoskeletal organization, and platelet production. When the gene is defective, multiple body systems are affected.
Clinical Characteristics of WAS
Patients with Wiskott-Aldrich syndrome typically experience a combination of:
- Immunodeficiency, leading to recurrent and severe infections
- Thrombocytopenia, resulting in increased bleeding and bruising
- Eczema and inflammatory skin disease
- Autoimmune complications
- Increased risk of malignancies
Symptoms often begin in infancy or early childhood and may worsen over time. Without definitive treatment, Wiskott-Aldrich syndrome can significantly reduce life expectancy and quality of life.
Traditional Treatment Approaches and Their Limitations
Historically, management of WAS involved a combination of supportive therapies, including:
- Infection prevention and treatment
- Immunoglobulin replacement therapy
- Platelet transfusions
- Management of autoimmune complications
While these measures help control symptoms, they do not address the underlying genetic cause.
Hematopoietic Stem Cell Transplantation (HSCT)
HSCT has been considered the only potentially curative option for Wiskott-Aldrich syndrome. However, its success depends heavily on the availability of a human leukocyte antigen (HLA)-matched related donor.
Challenges associated with HSCT include:
- Limited donor availability
- Risk of graft-versus-host disease
- Transplant-related complications
- Increased mortality in mismatched transplants
For patients without a suitable donor, treatment options were historically limited—until the development of etuvetidigene autotemcel.
What Is Waskyra ?
Waskyra, with the generic name etuvetidigene autotemcel, is a cell-based gene therapy designed to correct the genetic defect responsible for Wiskott-Aldrich syndrome.
Unlike donor-based transplantation, etuvetidigene autotemcel uses the patient’s own hematopoietic stem cells, which are genetically modified outside the body and then reintroduced.
This personalized approach eliminates the need for a matched donor while targeting the root cause of the disease.
FDA Approval of etuvetidigene autotemcel
The FDA approved Waskyra (etuvetidigene autotemcel) for:
- Pediatric patients aged six months and older
- Adult patients with Wiskott-Aldrich syndrome
Indicated Patient Population
etuvetidigene autotemcel is indicated for patients who meet all of the following criteria:
- Confirmed diagnosis of Wiskott-Aldrich syndrome
- Presence of a mutation in the WAS gene
- Considered appropriate candidates for HSCT
- No suitable HLA-matched related stem cell donor available
This approval represents a major regulatory milestone, reinforcing the clinical viability of gene therapy for rare immunodeficiency disorders.
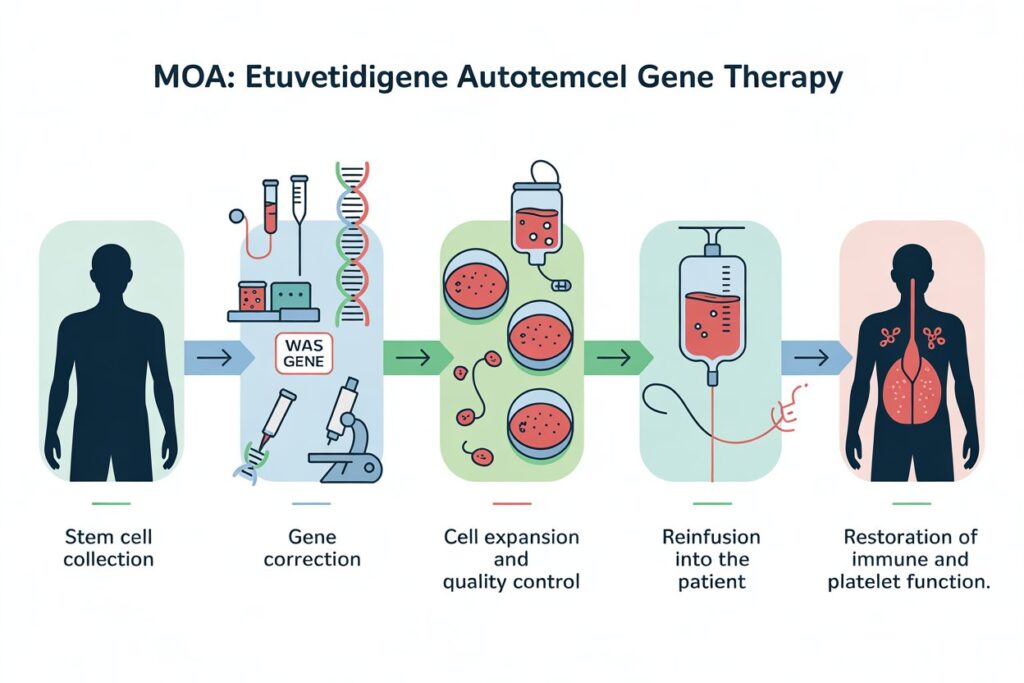
Mechanism of Action
The therapeutic effect of etuvetidigene autotemcel is achieved through genetic correction at the cellular level.
Step-by-Step Mechanism
- Stem Cell Collection
Hematopoietic stem cells are collected from the patient. - Gene Correction
A functional copy of the WAS gene is introduced into the collected stem cells using advanced gene-transfer technology. - Cell Expansion and Quality Control
The genetically corrected cells are expanded and undergo rigorous testing to ensure safety, viability, and gene expression. - Reinfusion into the Patient
The corrected stem cells are infused back into the patient, where they engraft in the bone marrow. - Restoration of Immune and Platelet Function
The corrected cells produce immune cells and platelets with functional WAS protein, improving immune response and clotting ability.
Through this mechanism, etuvetidigene autotemcel addresses the underlying cause of WAS rather than only managing symptoms.
Treatment Process and Clinical Administration
Treatment with etuvetidigene autotemcel is conducted in specialized medical centers experienced in gene therapy and stem cell transplantation.
Pre-Treatment Evaluation
Before therapy, patients undergo comprehensive assessments including:
- Genetic confirmation of WAS
- Immune system evaluation
- Organ function tests
- Infection screening
- Counseling for patients and caregivers
Conditioning Phase
Patients may receive conditioning therapy to prepare the bone marrow for the infusion of corrected stem cells. This phase is closely monitored to minimize complications.
Administration of etuvetidigene autotemcel
The corrected cells are administered intravenously in a controlled hospital setting. Patients are monitored for immediate reactions and early signs of engraftment.
Post-Treatment Monitoring
After infusion, patients undergo intensive follow-up to assess:
- Immune reconstitution
- Platelet recovery
- Infection frequency
- Overall clinical improvement
Safety Profile and Side Effects
As with any advanced cellular therapy, etuvetidigene autotemcel carries potential risks. Side effects may arise from conditioning regimens, immune recovery, or the infusion process itself.
Commonly Observed Side Effects
- Temporary low blood counts
- Increased susceptibility to infections during early recovery
- Infusion-related reactions
- Fatigue and weakness
Patients receiving etuvetidigene autotemcel are monitored continuously to detect and manage adverse effects promptly.
Drug Interactions and Precautions
Because etuvetidigene autotemcel affects the immune system and bone marrow, careful attention is required when managing concurrent medications.
Key Considerations
- Avoidance or careful timing of live vaccines
- Monitoring interactions with immunosuppressive drugs
- Adjustments to medications affecting bone marrow function
Healthcare providers tailor treatment plans to minimize risks and optimize outcomes.
Long-Term Follow-Up and Monitoring
Long-term follow-up is a critical component of treatment with etuvetidigene autotemcel. Patients may be monitored for several years to assess durability of response and overall health.
Follow-Up Evaluations Include
- Immune system function
- Platelet counts
- Autoimmune manifestations
- Growth and development in pediatric patients
- Quality of life assessments
This long-term data helps clinicians understand the sustained benefits and safety of etuvetidigene autotemcel.
Impact on Quality of Life
For many patients, etuvetidigene autotemcel offers the possibility of meaningful improvement in daily living.
Potential Benefits
- Reduced frequency of infections
- Improved bleeding control
- Decreased need for ongoing supportive therapies
- Enhanced physical activity and social participation
Families often report improved emotional well-being and reduced caregiving burden following successful treatment.
Ethical and Social Considerations
The approval of etuvetidigene autotemcel raises important discussions around access, affordability, and healthcare equity.
While gene therapy offers transformative benefits, ensuring equitable access remains a priority for healthcare systems worldwide.
Future Outlook
The success of etuvetidigene autotemcel sets the stage for broader application of gene therapies in rare genetic disorders.
Future directions may include:
- Earlier treatment intervention
- Expanded eligibility criteria
- Improved manufacturing efficiency
- Long-term outcome studies
This therapy represents a foundation upon which future gene-based treatments may build.
Conclusion
The FDA approval of Waskyra (etuvetidigene autotemcel) represents a milestone in the treatment of Wiskott-Aldrich syndrome. By offering a gene-corrective, patient-specific therapy for individuals without suitable donors, etuvetidigene autotemcel fills a long-standing gap in care.
As gene therapy continues to advance, etuvetidigene autotemcel stands as a powerful example of how precision medicine can transform outcomes for patients with rare, life-threatening genetic disorders

