Table of Contents
Introduction
Colorectal cancer remains one of the leading causes of cancer-related deaths worldwide. Over the past decade, advancements in targeted therapies have significantly improved survival outcomes for patients with specific genetic mutations. One of the most important developments in this field is the encorafenib and cetuximab FDA approval, which offers a targeted treatment option for patients with BRAF V600E-mutant metastatic colorectal cancer.
The encorafenib and cetuximab FDA approval marked a major milestone in oncology because it introduced a precision-based approach to treating patients whose tumors carry the BRAF mutation. Traditional chemotherapy treatments often provide limited effectiveness for these patients, and targeted therapies are designed to attack cancer cells more precisely.
This article explores everything you need to know about encorafenib and cetuximab FDA approval, including the mechanism of action, clinical trial evidence, safety profile, side effects, and drug interactions.
Understanding the Drugs: Encorafenib and Cetuximab
Before discussing the encorafenib and cetuximab FDA approval, it is important to understand the two drugs individually and how they work together.
Encorafenib
Encorafenib is an oral targeted therapy classified as a BRAF kinase inhibitor. It specifically blocks the mutated BRAF V600E protein that drives cancer cell growth.
BRAF mutations activate the MAPK signaling pathway, which causes uncontrolled cell proliferation. Encorafenib blocks this pathway and slows tumor progression.
Cetuximab
Cetuximab is a monoclonal antibody that targets the epidermal growth factor receptor (EGFR). It binds to the EGFR receptor on cancer cells and blocks signals that stimulate tumor growth.
When combined, these drugs create a dual-targeted therapy strategy, which is the basis of the encorafenib and cetuximab FDA approval.
Background Behind Encorafenib and Cetuximab FDA Approval
Patients with BRAF V600E-mutant metastatic colorectal cancer have historically had poor prognosis. Standard chemotherapy regimens offer limited effectiveness because the mutation activates multiple signaling pathways that help tumors resist treatment.
The encorafenib and cetuximab FDA approval was granted after studies showed that targeting both BRAF and EGFR pathways simultaneously improved treatment outcomes.
The combination therapy addresses tumor resistance mechanisms and improves survival compared with conventional treatments.
Because of these benefits, the encorafenib and cetuximab FDA approval quickly became an important advancement in precision oncology.. A more recent, expanded approval was granted on December 20, 2024, for use with chemotherapy (mFOLFOX6 or FOLFIRI), and this regimen received traditional approval on February 24, 2026
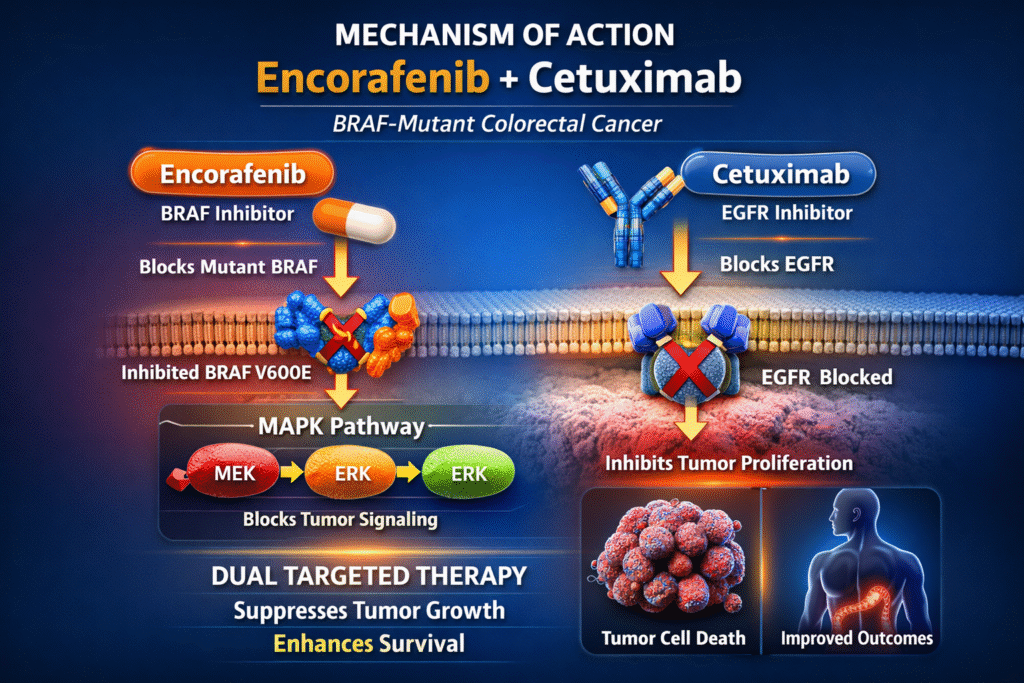
Mechanism of Action (MOA)
Understanding the molecular mechanism behind the encorafenib and cetuximab FDA approval helps explain why the combination therapy works better than single-agent treatments.
MAPK Pathway in Cancer
The MAPK signaling pathway regulates:
- Cell growth
- Cell differentiation
- Cell survival
In BRAF-mutant colorectal cancer, the BRAF protein becomes permanently activated.
This leads to:
- Continuous cell division
- Tumor progression
- Resistance to therapy
Encorafenib Mechanism
Encorafenib selectively inhibits the mutated BRAF V600E kinase.
This results in:
- Reduced MAPK pathway signaling
- Decreased tumor cell proliferation
- Increased cancer cell death
Cetuximab Mechanism
Cetuximab targets EGFR on cancer cells.
It blocks:
- EGFR ligand binding
- Receptor activation
- Downstream tumor signaling
Combined Mechanism
The success of the encorafenib and cetuximab FDA approval lies in the dual blockade strategy.
Encorafenib blocks the BRAF mutation while cetuximab prevents EGFR-mediated resistance pathways.
Together they:
- Suppress tumor growth
- Improve survival
- Reduce resistance
This combination therapy represents a major step forward in targeted cancer treatment.
Clinical Trials Supporting Encorafenib and Cetuximab FDA Approval
The encorafenib and cetuximab FDA approval was largely based on the results of a major clinical trial involving patients with metastatic colorectal cancer carrying the BRAF V600E mutation.
Study Design
The clinical trial evaluated:
- Encorafenib plus cetuximab
- Standard chemotherapy regimens
- Combination targeted therapies
Patients had previously received treatment but their disease had progressed.
Key Findings
The study showed that the combination therapy:
- Improved overall survival
- Increased response rates
- Reduced tumor progression
Patients receiving the treatment demonstrated significantly better outcomes compared with traditional therapy.
These results were critical in securing the encorafenib and cetuximab FDA approval.
Indications of Encorafenib and Cetuximab
Following the encorafenib and cetuximab FDA approval, the therapy is indicated for:
Treatment of Metastatic Colorectal Cancer
Specifically for patients with:
- BRAF V600E mutation
- Metastatic disease
- Prior therapy failure
Precision Oncology Approach
The encorafenib and cetuximab FDA approval highlights the importance of genetic testing before selecting therapy.
Patients must undergo molecular testing to confirm the presence of the BRAF mutation.
Dosage and Administration
After the encorafenib and cetuximab FDA approval, the recommended treatment regimen includes:
Encorafenib
- Oral capsule
- Taken once daily
Cetuximab
- Intravenous infusion
- Administered weekly or biweekly
Treatment continues until:
- Disease progression
- Unacceptable toxicity
Benefits of Encorafenib and Cetuximab Combination Therapy
The encorafenib and cetuximab FDA approval provides several clinical advantages.
Improved Survival
Patients experience longer survival compared with traditional chemotherapy.
Targeted Therapy
The treatment specifically targets mutated cancer cells.
Reduced Toxicity
Compared with aggressive chemotherapy regimens, targeted therapy may have a more manageable safety profile.
Precision Medicine Advancement
The encorafenib and cetuximab FDA approval represents progress toward personalized cancer treatment.
Side Effects of Encorafenib and Cetuximab
Although the encorafenib and cetuximab FDA approval demonstrated strong benefits, the treatment can still cause side effects.
Common Side Effects
Patients may experience:
- Fatigue
- Nausea
- Diarrhea
- Skin rash
- Abdominal pain
- Loss of appetite
- Joint pain
These side effects are generally manageable with supportive care.
Dermatologic Effects
Cetuximab often causes skin reactions due to EGFR inhibition.
Symptoms may include:
- Acne-like rash
- Dry skin
- Nail changes
These effects can indicate treatment activity.
Serious Side Effects
Less common but serious complications may include:
- Severe infusion reactions
- Cardiac complications
- Liver enzyme elevations
- Electrolyte imbalance
Doctors carefully monitor patients receiving therapy after the encorafenib and cetuximab FDA approval.
Drug Interactions
Drug interactions are an important consideration following the encorafenib and cetuximab FDA approval.
CYP Enzyme Interactions
Encorafenib is metabolized by liver enzymes.
Drugs that affect CYP enzymes may alter encorafenib levels.
Examples include:
- Certain antifungal drugs
- Some antibiotics
- Antiepileptic medications
Other Cancer Therapies
Combining additional targeted agents may increase toxicity.
Doctors evaluate each patient’s medication profile carefully.
Herbal Products
Herbal supplements such as St. John’s wort may reduce drug effectiveness.
Patients should inform healthcare providers about all medications they use.
Managing drug interactions is essential to ensure the safety and effectiveness of therapy after the encorafenib and cetuximab FDA approval.
Safety Monitoring
Patients receiving treatment under the encorafenib and cetuximab FDA approval require routine monitoring.
Blood Tests
Doctors monitor:
- Liver function
- Electrolyte levels
- Blood counts
Skin Monitoring
Dermatologic reactions should be evaluated regularly.
Cardiac Monitoring
Some patients may require ECG evaluation to detect cardiac effects.
Role of Biomarker Testing
The encorafenib and cetuximab FDA approval emphasizes the importance of biomarker-guided therapy.
BRAF Mutation Testing
Only patients with the BRAF V600E mutation benefit from the therapy.
Testing methods include:
- PCR-based assays
- Next-generation sequencing
Precision Medicine
Biomarker testing ensures that patients receive the most appropriate targeted treatment.
Impact on Oncology Practice
The encorafenib and cetuximab FDA approval significantly changed treatment strategies for metastatic colorectal cancer.
Before this therapy became available, patients with BRAF mutations had limited treatment options.
The combination therapy provides:
- Improved survival outcomes
- A targeted treatment strategy
- A new standard of care in specific cases
Many oncology guidelines now recommend this therapy as a key treatment option.
Future Research
Even after the encorafenib and cetuximab FDA approval, research continues to explore new treatment combinations.
Areas of investigation include:
Triple Combination Therapies
Researchers are studying combinations that include:
- BRAF inhibitors
- EGFR inhibitors
- MEK inhibitors
Earlier Use in Treatment
Clinical trials are evaluating whether the therapy could benefit patients earlier in the treatment process.
Combination with Immunotherapy
Some studies are exploring combinations with immune checkpoint inhibitors.
These advancements could further improve outcomes beyond the encorafenib and cetuximab FDA approval.
Conclusion
The encorafenib and cetuximab FDA approval represents a major breakthrough in the treatment of BRAF V600E-mutant metastatic colorectal cancer. By targeting both the BRAF mutation and EGFR signaling pathway, the combination therapy provides a more effective treatment strategy compared with traditional chemotherapy.
Patients receiving therapy after the encorafenib and cetuximab FDA approval benefit from improved survival, targeted cancer treatment, and a precision-based approach to oncology care. While side effects and drug interactions must be carefully monitored, the therapy has significantly expanded treatment options for patients with this aggressive form of cancer.
As research continues, the encorafenib and cetuximab FDA approval may pave the way for even more advanced targeted therapies and combination treatment strategies in the future.

