Table of Contents
Introduction
In the evolving field of oncology, hormonal therapy is an important part of breast cancer treatment. Among the latest advances is Elacestrant , an oral selective estrogen receptor degrader (SERD) that represents a significant leap forward in the management of estrogen receptor (ER)-positive, HER2-negative metastatic breast cancer.
Understanding the Elacestrant mechanism of action is important for clinicians, pharmacists, and researchers, as it provides insight into how this innovative therapy works at the molecular level to combat hormone-driven cancer progression.
In this comprehensive guide, we will explore what Elacestrant is, how it works, its pharmacological mechanism, clinical benefits, and its implications for the future of breast cancer therapy.
What Is Elacestrant?
Elacestrant (brand name Orserdu) is a next-generation, orally bioavailable selective estrogen receptor degrader (SERD) developed to overcome resistance to existing endocrine therapies in hormone receptor-positive breast cancer.
The US Food and Drug Administration (FDA) approved Elacestrant in January 2023 for the treatment of postmenopausal women or adult men with ER-positive, HER2-negative, ESR1-mutated advanced or metastatic breast cancer after disease progression after at least one line of endocrine therapy.
The approval marks a milestone — Elacestrant becomes the first oral SERD approved for clinical use, providing an alternative to traditional injectable SERDs such as fulvestrant (Faslodex).).
Why Is Elacestrant Important?
Hormone receptor-positive breast cancer accounts for about 70% of all breast cancer cases. For the past few decades, treatments have focused on the estrogen receptor (ER) pathway, aiming to block the increasing effects of estrogen on cancer cells.
However, long-term use of endocrine therapies such as tamoxifen (a selective estrogen receptor modulator, or SERM) and aromatase inhibitors (AIs) often leads to resistance.
Mutations in the ESR1 gene, which encodes estrogen receptor alpha (ERα), are the primary drivers of this resistance.
Elacestrant was specifically designed to target ESR1-mutated ERα, degrade it, and block its activity—even in the absence of estrogen binding inhibition.
In other words, Elacestrant MOA addresses one of the most challenging aspects of endocrine therapy resistance.
Elacestrant mechanism of action : Mechanism of Action Explained
To fully appreciate Elacestrant mechanism of action, it’s essential to understand how estrogen receptors function in breast cancer and how SERDs interfere with that process.
1. Role of Estrogen Receptors in Breast Cancer
- The estrogen receptor (ER) is a nuclear hormone receptor that, when bound to estrogen, translocates to the nucleus and binds to estrogen response elements (EREs) on DNA.
- This binding activates the transcription of genes that promote cell growth and proliferation.
- In ER-positive breast cancers, this pathway becomes hyperactive, leading to uncontrolled tumor growth.
2. How Traditional Therapies Work
- SERMs (like tamoxifen) compete with estrogen for receptor binding but act as partial agonists in some tissues.
- Aromatase inhibitors reduce estrogen production but do not affect receptor levels directly.
- Fulvestrant (an injectable SERD) binds to the receptor and promotes its degradation, but its poor bioavailability and injection route limit clinical flexibility.
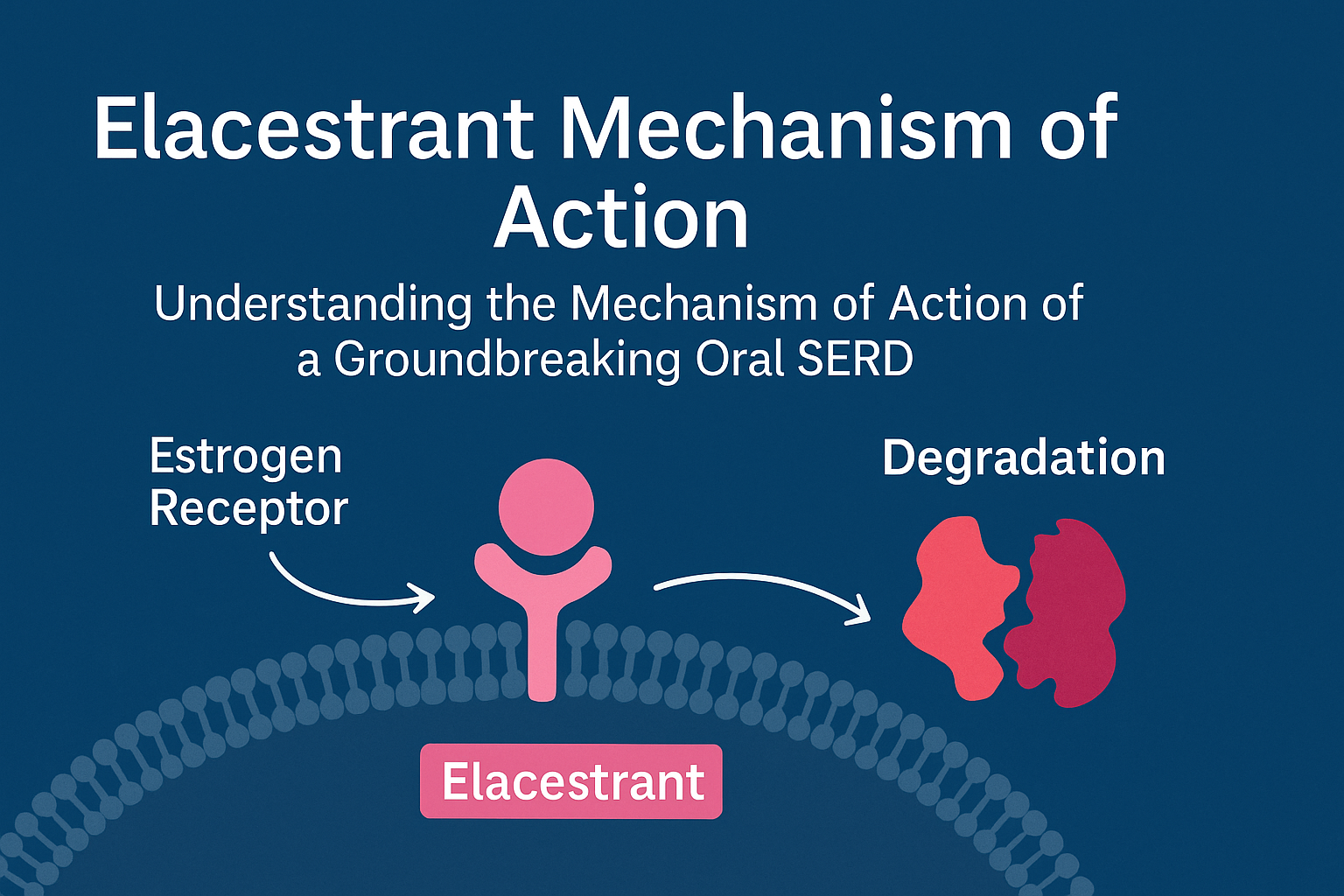
3. The Elacestrant Mechanism
Elacestrant mechanism of action combines high receptor binding affinity with potent receptor degradation activity, providing dual anti-estrogenic effects:
a. Binding to Estrogen Receptors
Elacestrant binds competitively to ERα, displacing endogenous estrogen. This prevents receptor activation and subsequent gene transcription.
b. Inducing Conformational Change
Upon binding, Elacestrant induces a conformational distortion in the receptor’s ligand-binding domain. This altered structure marks the receptor for ubiquitin-mediated degradation by the proteasome system.
c. Degradation of Estrogen Receptors
The receptor–Elacestrant complex is recognized by the cell’s protein degradation machinery, leading to irreversible receptor degradation and suppression of ER signaling.
d. Overcoming ESR1 Mutations
Elacestrant effectively targets mutated ESR1 variants (such as Y537S and D538G), which often confer resistance to other endocrine therapies.
By degrading the mutant receptor, Elacestrant prevents ligand-independent activation that drives tumor growth.
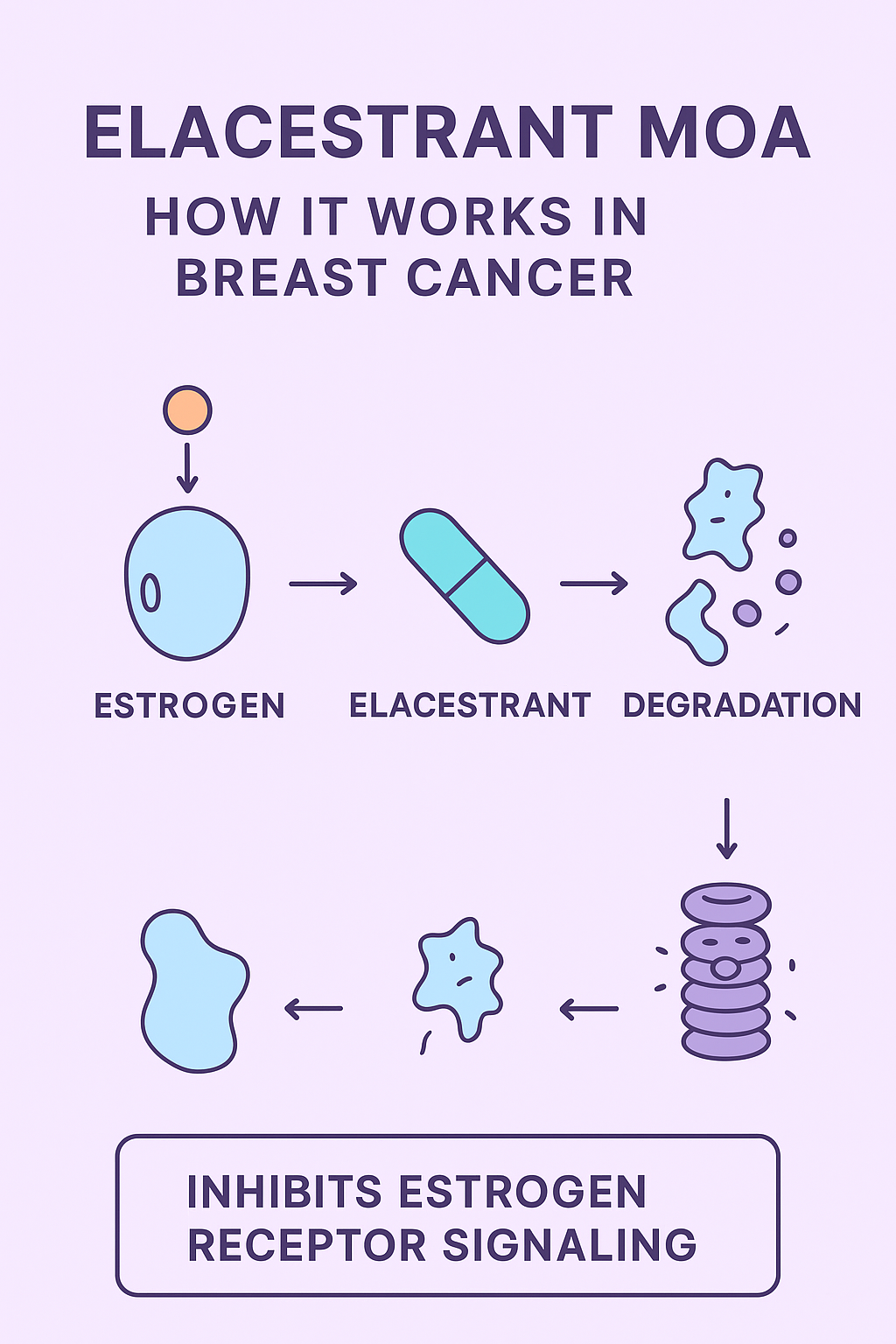
Elacestrant’s Pharmacodynamic and Pharmacokinetic Profile
Pharmacodynamics
• Elacestrant exhibits dose-dependent ER degradation in wild-type and mutant ESR1-positive cells.
• It reduces ER target gene expression, including progesterone receptor (PR) and cyclin D1, leading to a decrease in cell proliferation.
• Preclinical studies demonstrate robust anti-tumor efficacy compared to fulvestrant and other experimental SERDs.
Pharmacokinetics
- Route: Oral
- Bioavailability: High (unlike fulvestrant)
- Half-life: Approximately 30–35 hours
- Metabolism: Primarily via CYP3A4, with minor contributions from CYP2C9 and CYP2C19
- Excretion: Mainly fecal, minimal renal elimination
This favorable pharmacokinetic profile allows for once-daily oral dosing, improving patient adherence and convenience.
Elacestrant mechanism of action vs. Other SERDs
| Feature | Fulvestrant (Faslodex) | Elacestrant (Orserdu) |
| Route | Intramuscular injection | Oral tablet |
| Bioavailability | Low | High |
| ESR1 mutation activity | Partial | Strong |
| Onset of degradation | Slow | Rapid |
| Patient compliance | Moderate | Excellent |
| Combination potential | Limited | High |
Elacestrant mechanism of action thus provides a more complete and patient-friendly approach to estrogen receptor degradation.
Clinical Evidence Supporting Elacestrant mechanism of action
The EMERALD Phase III Trial was pivotal in validating Elacestrant’s clinical efficacy and its molecular mechanism.
Key Findings:
- Population: ER-positive, HER2-negative metastatic breast cancer patients who had progressed after prior endocrine therapy.
- Intervention: Elacestrant vs. standard-of-care endocrine therapy (fulvestrant or aromatase inhibitor).
- Results:
- Median progression-free survival (PFS): 3.8 months (Elacestrant) vs. 1.9 months (control).
- In ESR1-mutated subgroup: PFS reached 8.6 months with Elacestrant.
- Safety profile: Generally well tolerated; most common adverse effects were nausea and fatigue.
These outcomes confirm that Elacestrant mechanism of action effectively inhibits estrogen receptor–driven pathways, even in resistant disease settings.
Molecular Insights: How Elacestrant Targets ESR1 Mutations
Mutations in ESR1 lead to structural changes in ERα that allow receptor activation without estrogen.
Elacestrant uniquely binds to the altered receptor conformation and promotes allosteric destabilization, which:
- Prevents receptor dimerization.
- Reduces DNA-binding activity.
- Accelerates receptor turnover.
In doing so, it halts downstream signaling through PI3K/AKT and MAPK pathways — both critical for tumor proliferation and survival.
Potential Combination Therapies with Elacestrant
Given its mechanism, Elacestrant is being explored in combination with other targeted agents, including:
- CDK4/6 Inhibitors (e.g., Ribociclib, Palbociclib)
- Enhances anti-proliferative effects by simultaneously blocking ER signaling and cell cycle progression.
- PI3K/AKT/mTOR Pathway Inhibitors
- Addresses compensatory survival pathways activated during endocrine resistance.
- HER2-Targeted Agents
- For tumors exhibiting HER2-low expression in conjunction with ER positivity.
These combinations aim to extend the benefits of Elacestrant beyond monotherapy, especially in advanced and resistant settings.
Advantages of Elacestrant
- Oral administration – improves convenience and adherence.
- Effective against ESR1 mutations – overcomes a key resistance mechanism.
- Favorable safety profile – minimal severe toxicity.
- Versatile combination potential – fits well with emerging targeted therapies.
- Sustained receptor suppression – maintains efficacy over long-term dosing.
Adverse Effects and Safety Considerations
While Elacestrant is generally well tolerated, clinicians should be aware of potential side effects:
Common Adverse Effects
- Nausea
- Fatigue
- Arthralgia
- Vomiting
- Increased liver enzymes
Less Common Adverse Effects
- QT prolongation (rare)
- Hypercholesterolemia
Monitoring Parameters
- Baseline and periodic liver function tests
- Electrocardiogram (ECG) in patients with cardiac risk
- Routine lipid profile assessments
Elacestrant mechanism of action and the Future of Endocrine Therapy
The development of Elacestrant signals a paradigm shift in endocrine therapy.
Unlike earlier SERMs and SERDs, its oral availability, broad activity against ESR1 mutations, and robust degradation profile make it a leading candidate for next-generation ER targeting strategies.
Future studies are exploring Elacestrant in:
- Early-stage breast cancer (as adjuvant therapy).
- Combination regimens with CDK4/6 or PI3K inhibitors.
- Other estrogen-dependent malignancies, such as endometrial cancer.
By targeting the receptor directly rather than estrogen production, Elacestrant mechanism of action offers a more sustainable and precise anti-hormonal strategy.
Scientific Rationale Behind Elacestrant’s Success
Several structural and molecular features explain its clinical efficacy:
- High lipophilicity enables cell membrane permeability and efficient ER access.
- Conformational destabilization ensures complete receptor degradation.
- Selectivity for ERα minimizes off-target toxicity.
- Oral stability maintains consistent plasma concentrations for sustained activity.
These attributes, combined with its innovative design, position Elacestrant as a game-changer in endocrine therapy.
Conclusion
Understanding the Elacestrant mechanism of action is essential to appreciate its unprecedented role in the management of ER-positive, HER2-negative metastatic breast cancer. By downregulating both wild-type and mutant estrogen receptors, Elacestrant disrupts the very foundation of tumor growth signaling—offering hope for patients with endocrine-resistant disease.
As clinical trials expand its therapeutic potential, Elacestrant is poised to redefine how we approach hormone-driven cancers. With its oral delivery, potent receptor degradation, and favorable safety profile, it exemplifies the next evolution in targeted hormonal therapy.
Key Takeaways:
• Elacestrant is the first oral SERD approved for ER-positive, HER-2 negative, ESR-1 mutated metastatic breast cancer.
• The Elacestrant mechanism of action involves competitive receptor binding, conformational changes, and proteasomal degradation of ERα.
• It effectively targets ESR1 mutations that confer resistance to conventional therapies.
• Offers improved compliance, oral dosing convenience, and combination potential with other targeted agents.
• Represents a new frontier in endocrine therapy for breast cancer.
FAQs on Elacestrant mechanism of action