Table of Contents
Introduction
The recent Desmoda fda approval represents a major advancement in endocrine therapeutics, particularly in the treatment of central diabetes insipidus (CDI). DESMODA (desmopressin acetate) oral solution is now officially approved by the U.S. Food and Drug Administration for managing CDI in both adults and pediatric patients.
What makes this regulatory milestone particularly significant is that DESMODA is the first and only FDA-approved oral liquid formulation of desmopressin. For decades, desmopressin has been available in tablet, intranasal, and injectable forms. However, a standardized, commercially manufactured oral liquid option was lacking.
The Desmoda fda approval eliminates dependence on compounded liquid preparations, enhances dosing precision, and provides a safer and more controlled treatment alternative for vulnerable patient populations.
Understanding Central Diabetes Insipidus (CDI)
What Is Central Diabetes Insipidus?
Central diabetes insipidus is a rare disorder caused by insufficient secretion of antidiuretic hormone (ADH), also known as vasopressin, from the posterior pituitary gland.
Unlike diabetes mellitus, CDI is unrelated to blood sugar regulation. Instead, it involves abnormal water balance.
Hallmark Symptoms:
- Excessive urination (polyuria)
- Excessive thirst (polydipsia)
- Nocturia
- Dilute urine with low osmolality
- Risk of dehydration
- Electrolyte disturbances
Without treatment, CDI can result in serious complications such as hypernatremia and dehydration.
The Desmoda fda approval directly addresses the hormonal deficiency underlying CDI.
Causes of Central Diabetes Insipidus
CDI may develop due to:
- Pituitary surgery
- Traumatic brain injury
- Pituitary tumors
- Autoimmune disorders
- Genetic mutations
- Idiopathic causes
In all cases, vasopressin replacement therapy is required, making desmopressin the cornerstone of treatment.
What Is DESMODA?
DESMODA is an oral liquid formulation of desmopressin acetate, a synthetic analog of arginine vasopressin.
It selectively acts on renal V2 receptors to reduce urine output and restore fluid balance.
The Desmoda fda approval establishes DESMODA as the only FDA-approved liquid oral desmopressin product specifically indicated for CDI.
Why Desmoda FDA Approval Is Clinically Important
The Desmoda fda approval addresses several unmet clinical needs:
1. Standardization of Liquid Formulation
Previously, liquid desmopressin was often compounded in pharmacies. Compounded medications may vary in:
- Concentration
- Stability
- Accuracy
- Bioavailability
DESMODA provides consistent pharmaceutical-grade quality.
2. Pediatric Dosing Flexibility
Children often require precise titration. Tablets may not allow small incremental adjustments.
The Desmoda fda approval ensures:
- Accurate dose measurement
- Improved caregiver administration
- Safer pediatric treatment
3. Swallowing Difficulties in Adults
Many adult CDI patients struggle with tablets due to neurological conditions or post-surgical complications.
An oral solution simplifies administration.
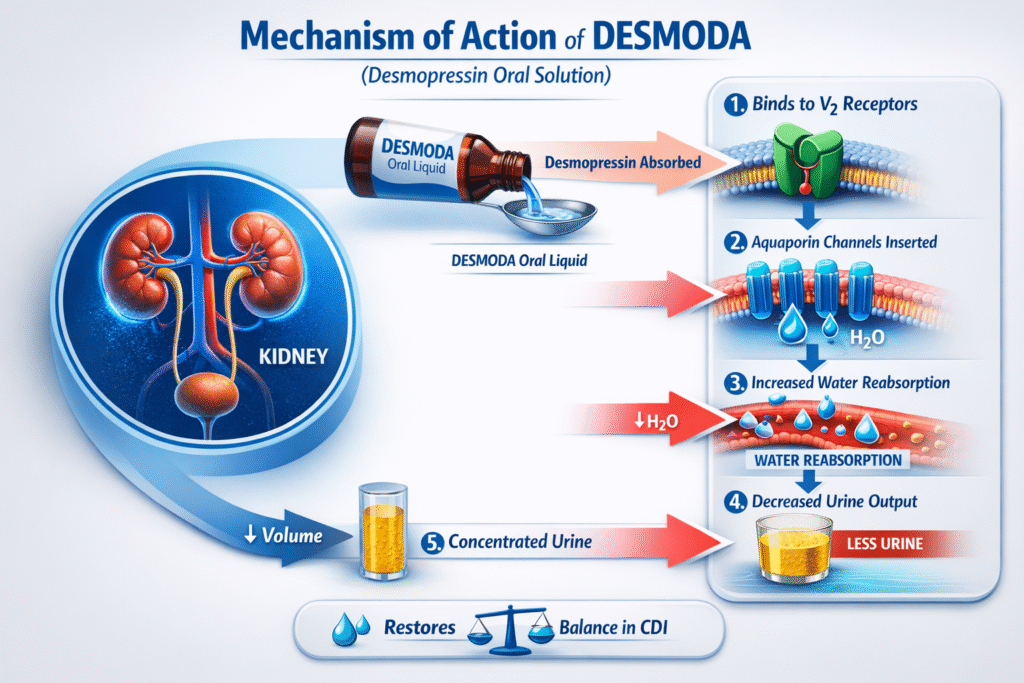
Mechanism of Action (MOA)
How DESMODA Works
Desmopressin selectively binds to vasopressin V2 receptors in the renal collecting ducts.
Mechanistic Steps:
- Activation of V2 receptors
- Stimulation of adenylate cyclase
- Increased cyclic AMP production
- Insertion of aquaporin-2 water channels
- Enhanced water reabsorption
- Reduced urine output
This mechanism restores plasma osmolality balance.
The scientific validation of this pathway was central to the Desmoda fda approval.
Pharmacokinetics
Pharmacokinetic evaluation during the Desmoda fda approval process showed:
- Predictable oral absorption
- Consistent bioavailability
- Controlled elimination
- Dose-proportional plasma concentrations
This consistency ensures safer sodium balance management.
Clinical Evidence Supporting Desmoda FDA Approval
The Desmoda fda approval was supported by:
Pharmacokinetic Bridging Studies
Compared oral solution to existing formulations.
Safety Evaluations
Confirmed known desmopressin safety profile.
Effectiveness Data
Demonstrated:
- Reduced urine output
- Increased urine osmolality
- Stable serum sodium (with monitoring)
Historical Clinical Experience
Desmopressin has decades of established clinical use, supporting the approval.
Indication
Following Desmoda fda approval, DESMODA is indicated for:
- Treatment of central diabetes insipidus
- Adult patients
- Pediatric patients
It is not indicated for nephrogenic diabetes insipidus.
Dosage and Administration
Dosing must be individualized.
General Guidelines:
- Initiate at lowest effective dose
- Adjust based on urine volume
- Monitor serum sodium regularly
- Restrict excessive fluid intake
Because desmopressin promotes water retention, inappropriate dosing can lead to dilutional hyponatremia.
The Desmoda fda approval includes strong guidance regarding sodium monitoring.
Side Effects
Common Side Effects
- Headache
- Nausea
- Abdominal discomfort
- Fatigue
- Mild fluid retention
Serious Side Effects
1. Hyponatremia
Most serious risk.
Symptoms:
- Confusion
- Seizures
- Severe headache
- Vomiting
- Altered mental status
2. Water Intoxication
3. Hypersensitivity Reactions (Rare)
Risk warnings were thoroughly evaluated before granting Desmoda fda approval.
Hyponatremia: The Key Safety Concern
Hyponatremia results from excessive water retention relative to sodium levels.
Risk Factors:
- High fluid intake
- Elderly patients
- Concomitant medications
- Renal impairment
Monitoring sodium at baseline and periodically is essential.
The safety framework was a central component of the Desmoda fda approval.
Drug Interactions
Drug interaction analysis was carefully reviewed during Desmoda fda approval.
Drugs That Increase Risk of Hyponatremia:
- SSRIs
- Tricyclic antidepressants
- NSAIDs
- Carbamazepine
- Thiazide diuretics
- Chlorpromazine
These drugs may enhance antidiuretic effects.
Drugs That May Reduce Effectiveness:
- Lithium
- Demeclocycline
These may interfere with renal response to desmopressin.
Clinicians should perform full medication reconciliation before prescribing.
Special Populations
Pediatric Use
The Desmoda fda approval includes children, making it highly valuable in pediatric endocrinology.
Geriatric Use
Higher hyponatremia risk.
Careful monitoring required.
Pregnancy
Use only if clearly needed.
Comparison With Other Desmopressin Forms
| Feature | Tablet | Nasal Spray | Injection | DESMODA |
| Oral Liquid | No | No | No | Yes |
| FDA Approved for CDI Liquid | No | No | No | Yes |
| Pediatric Titration | Limited | Variable | Hospital | Excellent |
The Desmoda fda approval sets a new benchmark for formulation precision.
Benefits of FDA-Approved Oral Liquid
The Desmoda fda approval offers:
- Pharmaceutical consistency
- Reduced compounding errors
- Improved caregiver confidence
- Simplified administration
- Better sodium management control
Post-Marketing Surveillance
After Desmoda fda approval, ongoing safety measures include:
- Adverse event reporting
- Hyponatremia surveillance
- Pediatric outcome tracking
- Real-world safety data collection
Risk-Benefit Assessment
Benefits:
- Effective CDI symptom control
- Accurate dosing
- Pediatric suitability
- Improved compliance
Risks:
- Hyponatremia
- Drug interactions
- Fluid imbalance
Regulators concluded that benefits outweigh risks when used according to labeling, leading to Desmoda fda approval.
Future Implications
The Desmoda fda approval may:
- Replace compounded oral desmopressin
- Improve endocrine treatment safety
- Enhance pediatric care standards
- Encourage more FDA-approved liquid endocrine therapies
Conclusion
The Desmoda fda approval represents a meaningful advancement in the treatment landscape for patients living with central diabetes insipidus. By introducing the first and only FDA-approved oral liquid formulation of desmopressin, this milestone addresses long-standing challenges related to dosing precision, compounding variability, and pediatric administration.
For adults and children with central diabetes insipidus, maintaining proper fluid balance is essential to prevent dehydration, electrolyte disturbances, and potentially life-threatening complications. The Desmoda fda approval ensures that patients now have access to a standardized, quality-controlled oral liquid option that delivers predictable pharmacokinetics and reliable therapeutic outcomes.
Clinically, this approval enhances:
- Accurate dose titration, especially in pediatric patients
- Safer management through clear sodium monitoring guidance
- Reduced dependence on pharmacy-compounded liquid preparations
- Improved convenience for patients with swallowing difficulties
While safety monitoring—particularly for hyponatremia—remains crucial, the overall benefit-risk profile evaluated during the Desmoda fda approval process confirms that DESMODA provides an effective and carefully regulated solution for CDI management.

