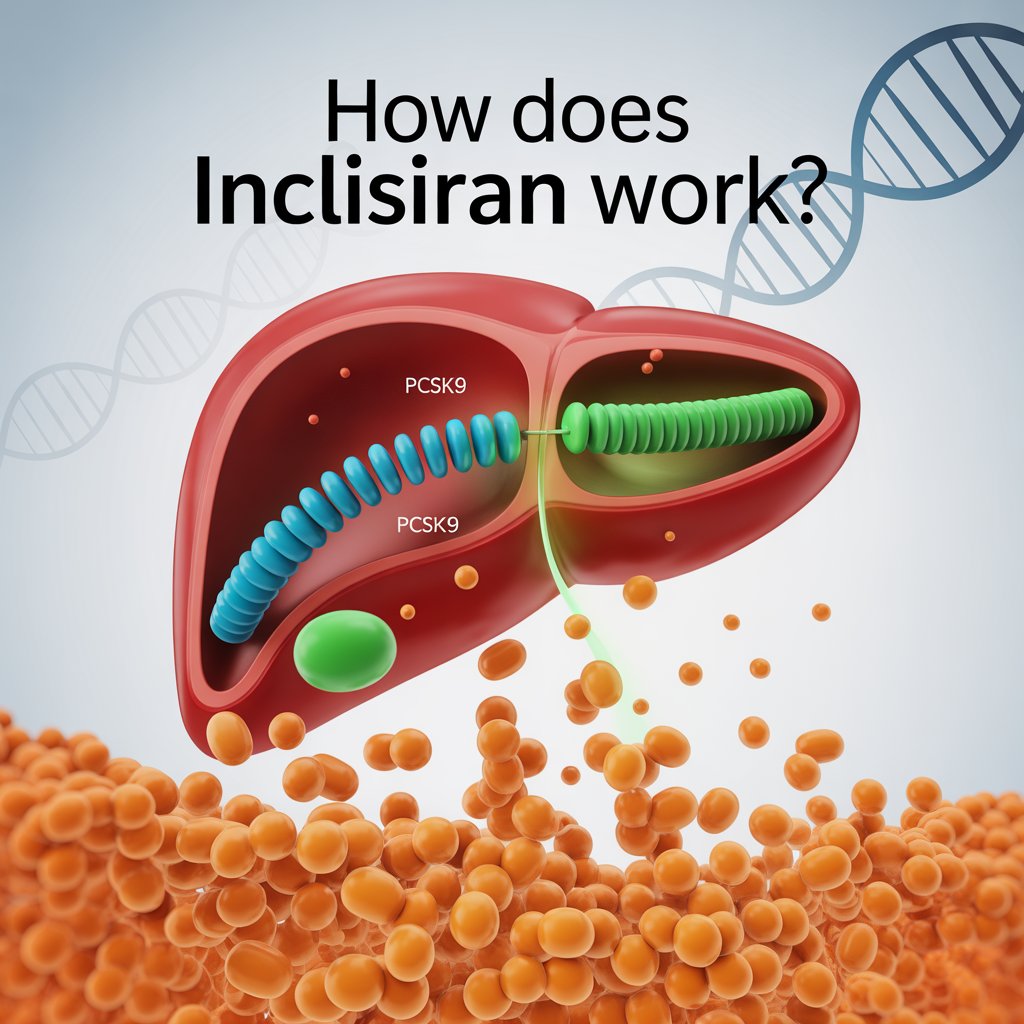
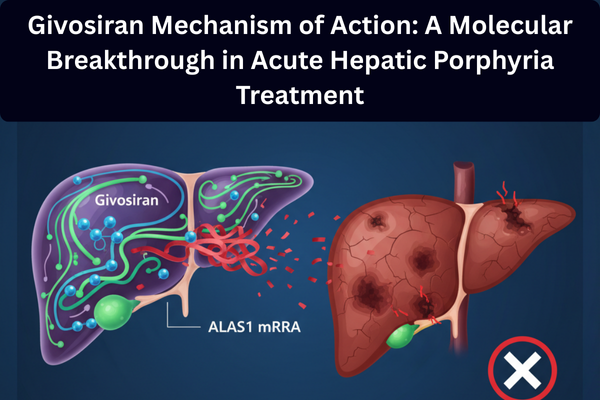
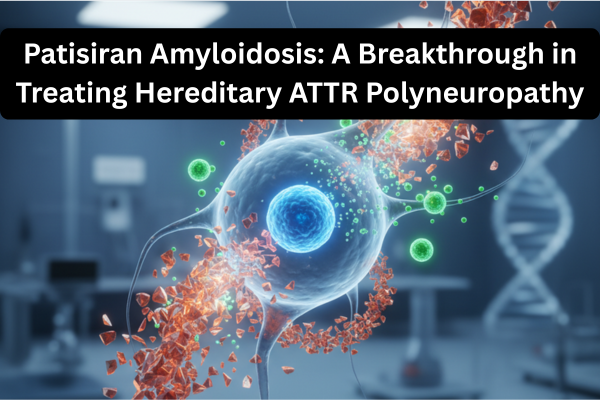
Nedosiran Approval 2025: Breakthrough RNAi Therapy for PH1
The nedosiran approval marks a significant milestone for patients suffering from Primary Hyperoxaluria Type 1 (PH1), a rare genetic disorder.
For individuals living with PH1, the accumulation of oxalate can lead to severe kidney damage and other systemic complications. The introduction of nedosiran approval as a treatment option brings new hope to these patients.