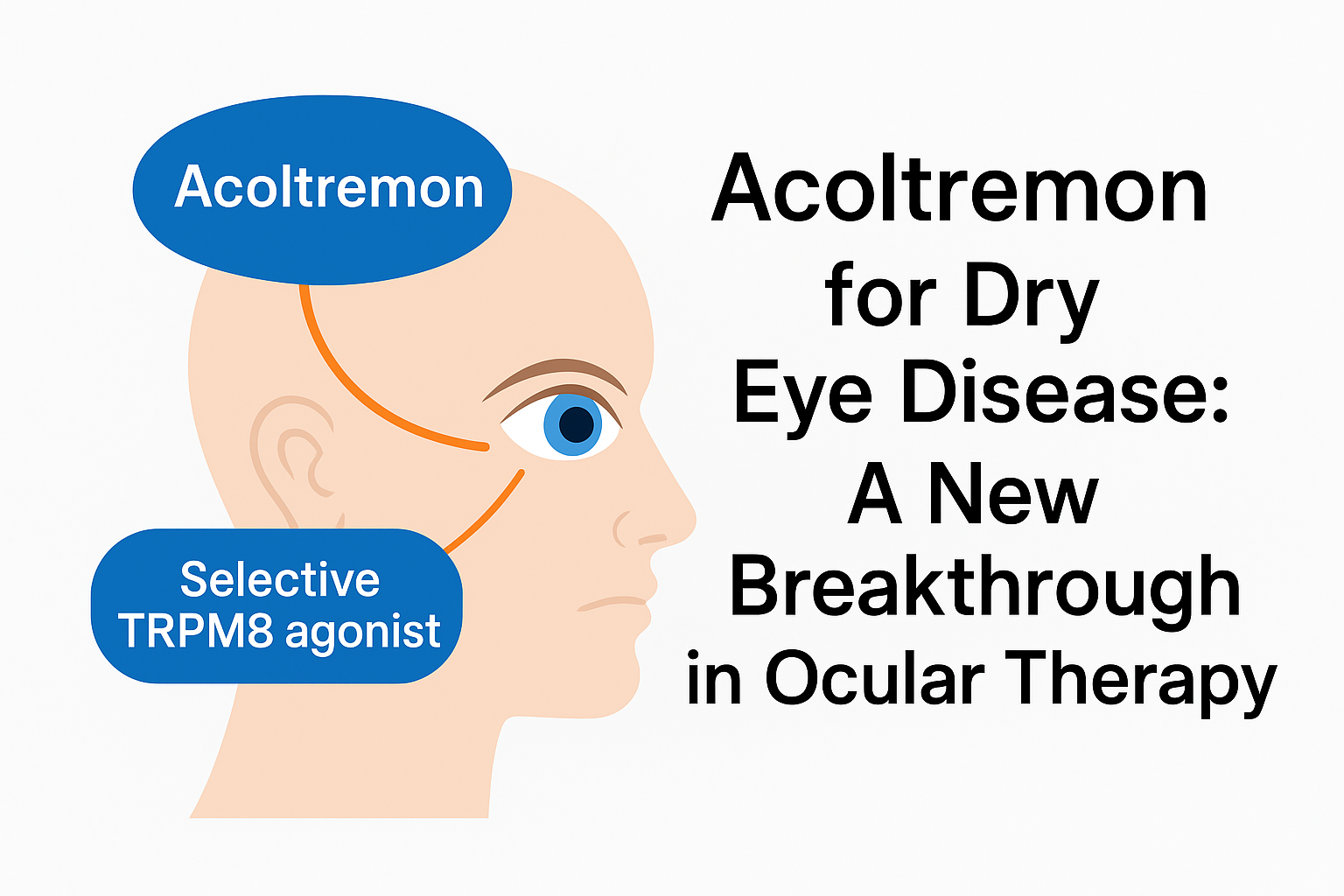
Berdazimer Gel: A Breakthrough Treatment for Molluscum Contagiosum (MC)
Introduction to Berdazimer Gel : What Is Berdazimer ? Berdazimer is a topical medication that was approved by the US FDA in 2024 for the treatment of molluscum contagiosum in patients 1 year of age and older. It is formulated at a 10.3% strength and is the first nitric oxide-releasing gel of its kind for …