Table of Contents
Introduction
Recent advances in precision medicine have significantly improved the treatment landscape for cancer. One notable development is the ojemda fda approval, which introduced a targeted therapy designed to treat certain pediatric brain tumors. Pediatric low-grade glioma (pLGG) is the most common brain tumor diagnosed in children, and although it often grows slowly, managing the disease can be challenging when tumors recur or fail to respond to standard therapy.
Traditional treatment strategies such as surgery, chemotherapy, and radiation therapy have been used for decades. While these methods can control tumor growth, they may also cause long-term complications, especially in children whose brains are still developing. As a result, researchers have focused on developing targeted treatments that act directly on the molecular drivers of cancer.
The ojemda fda approval represents a major step forward in this effort. The therapy targets specific genetic changes that activate tumor-growth pathways, allowing doctors to treat the disease more precisely. This approach not only improves treatment effectiveness but may also reduce exposure to toxic therapies.
In recent years, targeted drugs have become increasingly important in oncology. The ojemda fda approval reflects this shift toward personalized medicine, where treatments are selected based on the genetic characteristics of a tumor rather than using a one-size-fits-all approach.
For families affected by pediatric brain tumors, this approval offers renewed hope for improved disease management and better long-term outcomes.
What Is Ojemda?
Ojemda is an oral targeted therapy used to treat certain pediatric patients with low-grade glioma driven by abnormal signaling in the MAPK pathway. The active ingredient in the drug is tovorafenib, a kinase inhibitor designed to block specific proteins involved in tumor growth.
The therapy was developed to address tumors that contain BRAF gene alterations, such as BRAF fusions or rearrangements. These genetic abnormalities activate a signaling cascade that stimulates uncontrolled cell growth. By blocking this pathway, Ojemda helps slow tumor progression.
The ojemda fda approval allows physicians to prescribe the drug to pediatric patients who have relapsed or refractory disease after previous treatment. Because the medication targets a specific molecular pathway, it represents a more precise treatment option compared with conventional chemotherapy.
Ojemda is typically administered orally, making it easier for pediatric patients to receive treatment compared with intravenous therapies that require hospital visits.
Mechanism of Action (MOA)
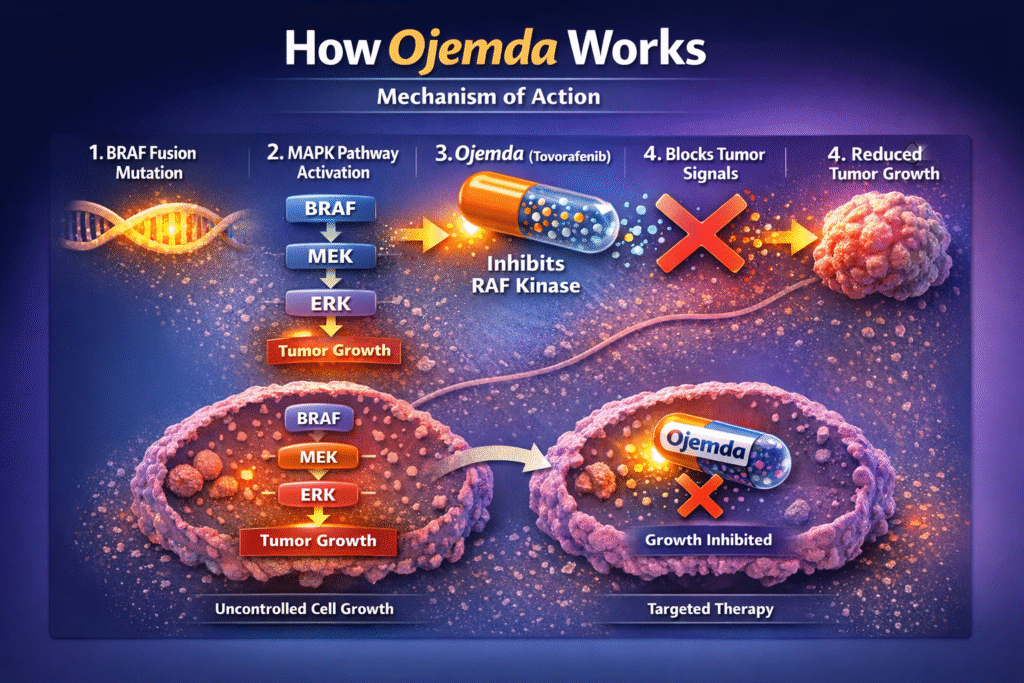
Understanding how Ojemda works helps explain why it is effective against certain tumors.
The drug functions as a type II RAF kinase inhibitor, meaning it blocks abnormal signaling within the MAPK pathway. This pathway plays a crucial role in regulating cell growth, survival, and division.
In many pediatric low-grade gliomas, mutations in the BRAF gene activate the MAPK signaling pathway. Once activated, the pathway continuously signals cells to grow and divide, leading to tumor formation.
Ojemda interferes with this process by inhibiting RAF kinases. By blocking these proteins, the drug interrupts the abnormal signaling that drives tumor growth.
Simplified Mechanism
- Genetic mutations activate the MAPK signaling pathway.
- The pathway sends continuous growth signals to tumor cells.
- Tovorafenib inhibits RAF kinases within the pathway.
- Abnormal signaling is reduced.
- Tumor growth slows or stabilizes.
Through this targeted mechanism, Ojemda helps control the progression of tumors associated with MAPK pathway activation.
Ojemda Fda Approval
The ojemda fda approval marked an important milestone in pediatric oncology.
On April 23, 2024, the U.S. Food and Drug Administration granted accelerated approval to tovorafenib (Ojemda), developed by Day One Biopharmaceuticals.
The approval covers the treatment of pediatric patients aged 6 months and older with relapsed or refractory low-grade glioma that has a BRAF fusion or rearrangement, and who require systemic therapy.
This regulatory decision was based on clinical trial results demonstrating meaningful tumor responses in children whose disease had progressed after previous treatment. Many patients experienced tumor shrinkage or disease stabilization, highlighting the potential of targeted therapy in this setting.
The accelerated approval pathway allows promising therapies to reach patients sooner when early clinical evidence suggests substantial benefit. Continued approval may depend on confirmatory studies that further demonstrate clinical effectiveness.
For physicians treating pediatric brain tumors, the ojemda fda approval provides an additional treatment option for patients with limited alternatives.
Clinical Benefits
The introduction of Ojemda brings several advantages for pediatric cancer care.
Key Benefits
• Targeted therapy based on tumor genetics
• Oral administration for easier treatment
• Potential reduction in chemotherapy exposure
• Improved disease control in some patients
• Personalized treatment strategy
Because the therapy specifically targets MAPK pathway activation, it may be particularly effective in tumors driven by BRAF alterations.
The ojemda fda approval therefore represents an important advancement in precision oncology.
Dosage and Administration of Ojemda Fda Approval
Ojemda is administered orally and dosing may depend on factors such as body weight and age.
Doctors typically prescribe the medication based on established treatment guidelines. Patients receiving the therapy should be monitored regularly to ensure optimal safety and effectiveness.
Routine monitoring may include:
• Blood tests
• Liver function assessments
• Physical examinations
• Imaging studies to evaluate tumor response
Healthcare providers may adjust the dose if adverse reactions occur or if patients experience difficulty tolerating the medication.
Side Effects
Like most cancer therapies, Ojemda can cause side effects. However, many adverse reactions observed during clinical studiesof Ojemda Fda Approval and were manageable with supportive care or dose adjustments.
Common Side Effects
• Rash
• Hair color changes
• Fatigue
• Headache
• Nausea
• Vomiting
Less Common but Serious Effects
• Liver enzyme elevation
• Muscle enzyme increases
• Skin reactions
• Eye disorders
Because pediatric patients may respond differently to treatment, doctors carefully monitor them throughout therapy.
Early detection and management of side effects help ensure that treatment remains safe and effective.
Drug Interactions
Drug interactions are an important consideration when prescribing targeted therapies.
Certain medications may alter how Ojemda is metabolized in the body, potentially increasing toxicity or reducing effectiveness.
Possible Interacting Drugs
• Strong CYP enzyme inhibitors
• CYP enzyme inducers
• Certain antifungal medications
• Some antibiotics
• Antiepileptic drugs
Patients should inform their healthcare provider about all medications they are taking, including over-the-counter drugs and herbal supplements.
Proper medication review helps prevent unwanted interactions during treatment.
Monitoring and Safety Considerations
Because Ojemda is a targeted anticancer therapy, regular monitoring is recommended during treatment.
Healthcare providers may perform the following assessments:
• Liver function testing
• Blood chemistry monitoring
• Eye examinations
• Neurological evaluation
These tests help detect potential complications early and ensure that the therapy continues to provide clinical benefit.
Long-term safety monitoring is also important, particularly in pediatric populations receiving extended treatment.
Future Outlook
The approval of targeted therapies such as Ojemda highlights the growing importance of molecular-based treatment strategies in oncology.
Researchers continue to explore additional uses for RAF inhibitors in other cancers driven by MAPK pathway activation. Ongoing clinical trials may expand the potential applications of this therapy in the future.
Advances in genomic testing are also making it easier to identify patients who may benefit from targeted treatment.
The ojemda fda approval therefore represents not only a new therapy but also a broader shift toward personalized cancer care.
Conclusion
The ojemda fda approval represents a significant advancement in the treatment of pediatric low-grade glioma. By targeting abnormal MAPK signaling caused by BRAF alterations, this therapy offers a more precise approach to controlling tumor growth.
With its oral administration, targeted mechanism, and promising clinical results, Ojemda provides an important new option for pediatric patients whose disease has progressed after earlier treatments.
As research continues, targeted therapies like this one are expected to play an increasingly important role in improving outcomes for children with brain tumors.

