Table of Contents
Introduction
Respiratory viral infections continue to pose a serious health threat worldwide, particularly among older adults. Seasonal influenza and COVID-19 caused by SARS-CoV-2 remain two of the most significant infectious diseases affecting people over the age of 50. Healthcare systems globally are constantly seeking new strategies to protect vulnerable populations from these illnesses.
One innovative solution is mCombriax, a modern vaccine designed to provide protection against both influenza and COVID-19. This medical advancement represents an important step toward simplifying vaccination strategies while maintaining strong immune protection.
Unlike traditional vaccination programs that require separate injections for flu and COVID-19 prevention,the vaccine combines protection against both viruses into a single immunization. This approach not only improves convenience but may also increase vaccination compliance among older adults.
As individuals age, their immune systems naturally become weaker. For this reason, vaccines play a crucial role in maintaining public health and preventing severe disease. The development of this vaccine demonstrates how modern immunology and biotechnology are working together to address global health challenges.
In this comprehensive guide, we will explore the key clinical and scientific aspects of mCombriax, including its mechanism of action, regulatory approval pathway, safety profile, side effects, and possible drug interactions.
What is mCombriax?
mCombriax is a combination vaccine designed for active immunisation against influenza and COVID-19 caused by the SARS-CoV-2 virus.
This immunization is specifically indicated for individuals aged 50 years and older, who are at higher risk of complications from respiratory infections.
The vaccine introduces viral components that stimulate the immune system without causing disease. Once administered, the vaccine prepares the body to recognize and defend itself against both influenza viruses and SARS-CoV-2.
A key advantage of this vaccine is its ability to provide dual protection through a single vaccination strategy. This reduces the need for multiple injections during respiratory virus seasons.
The introduction of mCombriax reflects the growing trend toward combination vaccines that improve vaccination coverage while maintaining strong safety standards.
Importance of Preventing Influenza and COVID-19
Influenza and COVID-19 can cause severe illness in older adults. Complications from these infections may include pneumonia, hospitalization, and long-term health problems.
Vaccination remains the most effective preventive measure against these diseases. By stimulating the immune system, vaccines help reduce the risk of severe illness and viral spread.
The development of the vaccine offers a convenient preventive approach by addressing two major respiratory threats simultaneously.
Public health experts believe that combination vaccines like mCombriax could significantly improve vaccination rates among adults who may otherwise skip recommended immunizations.
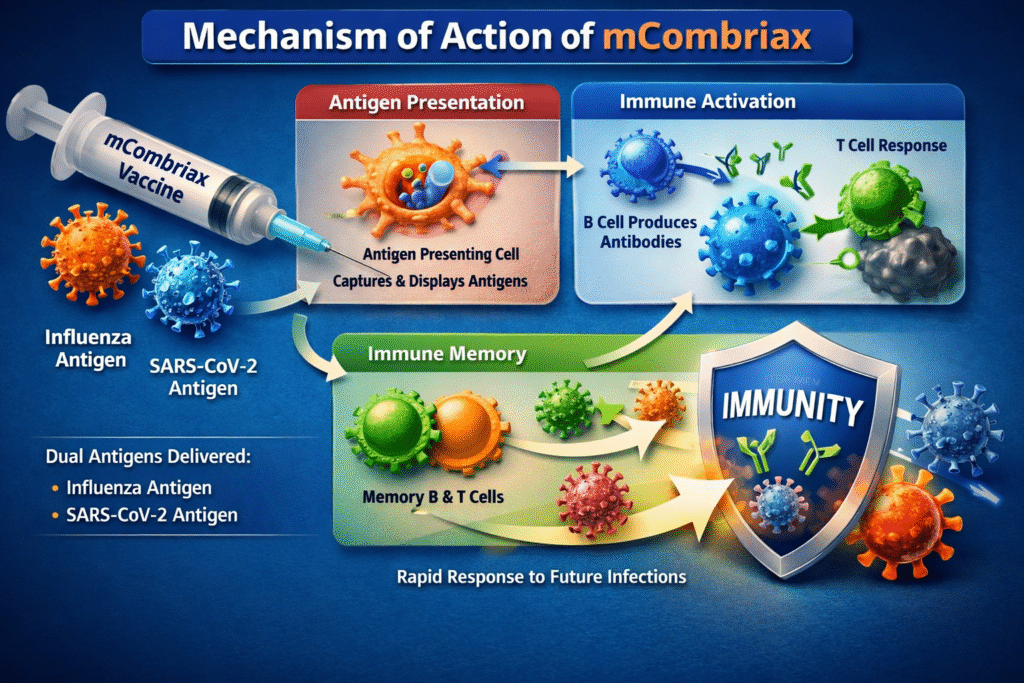
Mechanism of Action
The mechanism of action is based on the fundamental principles of immunology. After administration, the vaccine introduces specific viral antigens that train the immune system to recognize harmful pathogens.
Antigen Recognition
Once mCombriax is injected into the body, immune cells called antigen-presenting cells detect and process the viral antigens contained in the vaccine.
These cells then present the antigens to other immune cells, initiating the immune response.
Antibody Production
The immune system responds by producing antibodies specifically designed to recognize influenza viruses and the SARS-CoV-2 virus.
These antibodies remain in circulation and provide protective immunity.
Immune Memory
Another important feature ofthe vaccine is the development of immune memory. Memory B-cells and T-cells remember the viral antigens, allowing the body to respond rapidly if exposure occurs in the future.
Because of this mechanism, mCombriax helps prevent severe disease and complications associated with respiratory viral infections.
FDA Approval and Regulatory Evaluation
All vaccines must undergo extensive testing before being approved for use in the general population.
The development process for mCombriax involved several phases of clinical research designed to ensure safety and effectiveness.
Preclinical Studies
Before human testing began, laboratory research evaluated the biological activity and safety profile of the vaccine.
Clinical Trials
The clinical development of this vaccine included multiple trial phases.
Phase I Trials
Researchers tested mCombriax in small groups to assess safety and dosage levels.
Phase II Trials
The vaccine’s ability to generate immune responses was studied in a larger population.
Phase III Trials
Large clinical trials evaluated the effectiveness of the vaccine in preventing influenza and COVID-19 infections.
These trials provided the scientific evidence needed for regulatory review.
Dosage and Administration
The vaccination is administered as an intramuscular injection by trained healthcare professionals.
Typically, mCombriax is injected into the upper arm muscle. The exact vaccination schedule may vary depending on public health recommendations.
Healthcare providers determine when this vaccine should be given during seasonal vaccination campaigns.
Because the vaccine provides dual protection, mCombriax may help simplify immunization schedules for older adults.
Side Effects
Like all vaccines, mCombriax may cause mild side effects as the immune system responds to the immunization.
Common Side Effects
The most frequently reported reactions include:
- Injection site pain
- Fatigue
- Headache
- Muscle aches
- Mild fever
These symptoms usually disappear within a few days.
Moderate Reactions
Some individuals receiving :
- Chills
- Joint pain
- Temporary weakness
- Swelling at the injection site
These reactions are generally short-lived.
Rare Adverse Effects
Serious reactions are rare but may include allergic responses or hypersensitivity reactions.
Patients experiencing severe symptoms after receiving mCombriax should seek immediate medical care.
Drug Interactions
Vaccines typically have fewer interactions compared with pharmaceutical drugs. However, certain medications may influence immune response.
Immunosuppressive Therapy
Medications that suppress the immune system may reduce the effectiveness of mCombriax.
These drugs may include corticosteroids or chemotherapy agents.
Co-administration with Other Vaccines
In some cases, mCombriax may be administered alongside other vaccines, depending on healthcare guidelines.
Chronic Disease Medications
Most medications used for chronic conditions do not interfere with mCombriax, but healthcare providers should always review a patient’s medication list before vaccination.
Safety and Precautions
Before receiving this vaccine , patients should discuss their medical history with a healthcare provider.
Precautions include:
- History of severe allergic reactions
- Immunocompromised conditions
- Current illness or fever
Healthcare providers evaluate whether mCombriax is appropriate for each patient based on individual health status.
Public Health Significance
Combination vaccines represent a major advancement in preventive medicine. By offering protection against multiple pathogens, they simplify vaccination programs and increase coverage.
The introduction of mCombriax demonstrates how vaccine innovation can address multiple health challenges simultaneously.
As respiratory viruses continue to evolve, preventive strategies such as mCombriax may become increasingly important in protecting vulnerable populations.
Conclusion
The development of mCombriax marks a significant milestone in modern vaccine science. By combining protection against influenza and COVID-19 into a single immunization, this vaccine offers a convenient and efficient preventive strategy for adults aged 50 years and older.
Through its immune-stimulating mechanism, this vaccine prepares the body to recognize and fight viral infections before they cause severe illness.
Clinical research and regulatory evaluations ensure that the vaccine meets strict safety and effectiveness standards before reaching patients.
As vaccination technology continues to evolve, combination vaccines like mCombriax may play an increasingly important role in global disease prevention and public health protection.

