Table of Contents
Introduction
Pegzilarginase-nbln is an innovative enzyme replacement therapy designed to treat patients suffering from arginase 1 deficiency, a rare genetic disorder that disrupts the urea cycle. This therapy has attracted significant attention in the medical and pharmaceutical communities because it directly targets the underlying metabolic imbalance responsible for the disease.
Arginase deficiency leads to elevated arginine levels in the blood, causing progressive neurological complications, muscle stiffness, and developmental delays. Traditional treatments for this disorder have focused primarily on dietary restrictions and supportive therapies. However, these approaches often fail to adequately control arginine levels.
Pegzilarginase-nbln represents a promising breakthrough because it offers a targeted biochemical solution. By replacing the missing enzyme activity, Pegzilarginase-nbln can help restore metabolic balance and reduce toxic arginine accumulation.
Over the past few years, Pegzilarginase-nbln has undergone extensive clinical trials evaluating its safety, efficacy, and pharmacological profile. These studies suggest that Pegzilarginase-nbln may significantly improve outcomes for patients with arginase deficiency.
This comprehensive article explores everything you need to know about Pegzilarginase-nbln, including its mechanism of action, side effects, FDA approval status, drug interactions, clinical benefits, and future prospects.
What is Pegzilarginase-nbln (Loargys)?
Pegzilarginase-nbln is a pegylated recombinant human arginase enzyme therapy developed to treat arginase 1 deficiency (ARG1-D).
Arginase deficiency is a rare urea cycle disorder (UCD) characterized by impaired conversion of arginine into ornithine and urea. This leads to excessive arginine accumulation in the bloodstream.
Pegzilarginase-nbln works by replacing the missing enzyme activity and restoring the normal metabolic pathway.
Key characteristics of Pegzilarginase-nbln
• Recombinant enzyme therapy
• Pegylated formulation for extended circulation time
• Targets arginine metabolism
• Designed for long-term treatment of ARG1 deficiency
• Developed for both pediatric and adult patients
The development of Pegzilarginase-nbln represents a major advancement in precision metabolic medicine.
Understanding Arginase 1 Deficiency
Arginase 1 deficiency is caused by mutations in the ARG1 gene, which produces the arginase enzyme responsible for breaking down arginine in the liver.
When this enzyme is deficient or nonfunctional, arginine accumulates in the body.
Common symptoms of arginase deficiency
• Progressive spasticity
• Developmental delay
• Intellectual disability
• Seizures
• Growth problems
• Elevated blood arginine levels
Without proper treatment, patients may experience severe neurological complications.
Pegzilarginase-nbln aims to correct the metabolic imbalance responsible for these symptoms.
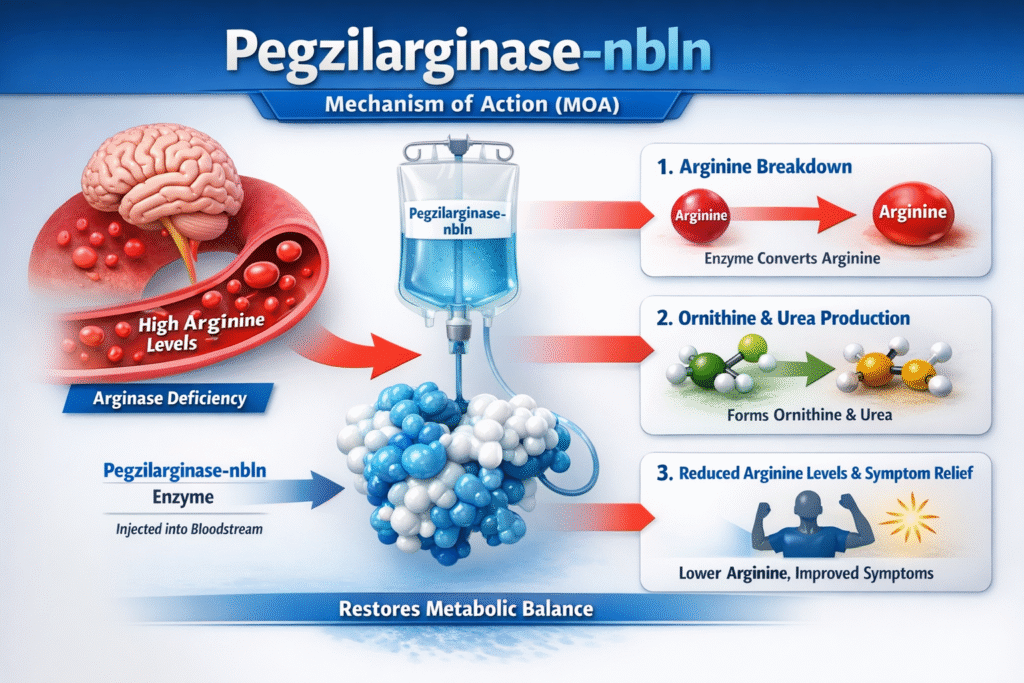
Mechanism of Action (MOA)
The Pegzilarginase-nbln mechanism of action focuses on reducing excess arginine in the bloodstream.
Normally, arginase converts arginine into ornithine and urea. In arginase deficiency, this step is impaired.
Pegzilarginase-nbln introduces an active enzyme that performs the same metabolic function.
Step-by-step MOA
- Pegzilarginase-nbln circulates in the bloodstream after administration.
- The recombinant arginase enzyme catalyzes the breakdown of arginine.
- Arginine is converted into ornithine and urea.
- Blood arginine levels gradually decrease.
- Reduced arginine levels improve neurological symptoms.
Why Pegylation Matters
Pegzilarginase-nbln contains a polyethylene glycol (PEG) modification, which offers several advantages:
• Increased half-life
• Improved enzyme stability
• Reduced immune response
• Less frequent dosing
This advanced design helps Pegzilarginase-nbln remain active in the bloodstream for longer periods.
Pharmacokinetics
Understanding the pharmacokinetics of Pegzilarginase-nbln is essential for clinicians and pharmacists.
Absorption
It is typically administered via intravenous infusion, allowing direct entry into systemic circulation.
Distribution
After administration, it circulates throughout the bloodstream where it interacts with arginine molecules.
Metabolism
The enzyme gradually breaks down arginine into urea and ornithine.
Elimination
The pegylated enzyme is eventually metabolized and cleared through normal protein degradation pathways.
Clinical Trials of Loargys
Clinical trials have played a crucial role in demonstrating the therapeutic potential of Pegzilarginase nbln.
These studies focused on patients with arginase 1 deficiency and evaluated several clinical endpoints.
Key outcomes from clinical trials
• Significant reduction in plasma arginine levels
• Improvement in mobility and motor function
• Reduction in neurological complications
• Acceptable safety profile
Many patients receiving Pegzilarginase-nbln showed sustained metabolic improvement during long-term therapy.
These promising results support the continued development and regulatory review of Pegzilarginase-nbln.
Pegzilarginase-nbln FDA Approval Status
The FDA approval process for Pegzilarginase nbln has been closely monitored by the pharmaceutical communit.It is approved by the FDA in February 2026 for treating Arginase-1 Deficiency (ARG1-D) in patients aged 2 and older.
Because arginase deficiency is an extremely rare disease, Loargys received Orphan Drug designation.
This designation helps accelerate the development of therapies for rare conditions.
Regulatory highlights
• Investigational therapy for arginase deficiency
• Evaluated in multiple clinical trials
• Demonstrated reduction in plasma arginine levels
• Potential disease-modifying therapy
The progress toward regulatory approval highlights the importance of Loargys in treating rare metabolic disorders.
Benefits of Therapy
Loargys offers several advantages compared to conventional treatment strategies.
Major therapeutic benefits
1. Targeted enzyme replacement
Pegzilarginase-nbln directly replaces the missing enzyme activity.
2. Reduced arginine levels
Lower plasma arginine levels reduce toxicity and neurological damage.
3. Improved patient mobility
Some clinical trial participants showed improved motor function.
4. Long-acting formulation
The pegylated design allows prolonged enzyme activity.
5. Disease-modifying potential
Pegzilarginase-nbln addresses the root cause of the disorder rather than just symptoms.
Loargys Side Effects
However, clinical trials suggest that most adverse effects are manageable.
Common side effects
• Headache
• Nausea
• Fatigue
• Injection site reactions
• Mild gastrointestinal discomfort
Less common side effects
• Hypersensitivity reactions
• Elevated liver enzymes
• Allergic responses
Serious adverse reactions
Although rare, serious reactions may include:
• Severe hypersensitivity
• Immune-mediated reactions
Patients receiving Pegzilarginase nbln should be monitored regularly by healthcare professionals.
Drug Interactions with Loargys
Understanding drug interactions is essential when prescribing Pegzilarginase nbln.
Currently, limited drug interaction data is available because the therapy is relatively new.
However, several theoretical interactions should be considered.
Possible interaction categories
1. Urea cycle medications
Drugs used to treat urea cycle disorders may interact with Loargys metabolic pathways.
2. Immunomodulators
Medications that affect immune function could influence enzyme therapy response.
3. Protein-based therapies
Concurrent biologic therapies may increase the risk of immune reactions.
Healthcare providers should evaluate patient medication history before initiating Pegzilarginase-nbln.
Dosage and Administration
Loargys is administered under medical supervision.
Typical administration method
• Intravenous infusion
• Given at scheduled intervals
• Dose adjusted based on arginine levels
Regular monitoring of plasma arginine concentrations is recommended during therapy.
Safety Monitoring During Treatment
Patients receiving Loargys require ongoing monitoring.
Recommended monitoring parameters
• Plasma arginine levels
• Liver function tests
• Neurological assessment
• Hypersensitivity reactions
Routine follow-up ensures the therapy remains safe and effective.
Future Potential of Loargys
Pegzilarginase nbln may represent the future of metabolic disease therapy.
Researchers are continuing to study its long-term benefits.
Potential future applications include:
• Early treatment in pediatric patients
• Combination therapy with dietary management
• Expanded use in other arginine-related disorders
If future trials continue to show positive results, Pegzilarginase-nbln could become the standard of care for arginase deficiency.
Role of Loargys in Rare Disease Treatment
Rare diseases often lack effective treatments due to limited research funding and small patient populations.
Pegzilarginase nbln demonstrates how modern biotechnology can address these challenges.
Biologic enzyme therapies like Pegzilarginase-nbln are transforming the treatment landscape for rare metabolic conditions.
Conclusion
Pegzilarginase-nbln represents a significant advancement in the treatment of arginase 1 deficiency, a rare but serious metabolic disorder. By replacing the missing enzyme responsible for arginine metabolism, Loargys directly addresses the underlying cause of the disease.
Clinical trials have demonstrated promising results, including substantial reductions in plasma arginine levels and improvements in patient neurological outcomes. The pegylated enzyme design also enhances the drug’s stability and circulation time, making Loargys a powerful therapeutic option.
While ongoing research continues to evaluate its long-term safety and effectiveness, Pegzilarginase nbln has already generated considerable excitement among clinicians, researchers, and patients.
As regulatory evaluations progress and additional clinical data become available, Pegzilarginase nbln may soon become a cornerstone therapy for individuals living with arginase deficiency.
The development of Pegzilarginase-nbln highlights the growing role of biotechnology and enzyme replacement therapies in modern medicine.

