Table of Contents
Introduction
The difamilast FDA approval marks an important milestone in dermatology and inflammatory skin disease treatment. Atopic dermatitis is one of the most common chronic inflammatory skin disorders worldwide, affecting millions of children and adults. Patients often experience itching, redness, inflammation, and recurring flare-ups that significantly impact their quality of life.
Traditional therapies such as topical corticosteroids and calcineurin inhibitors have been the primary treatment options for many years. However, these medications may cause long-term side effects, including skin thinning or irritation. As a result, researchers have been working to develop safer non-steroidal treatments for chronic inflammatory skin conditions.
The difamilast FDA approval introduces a promising non-steroidal topical therapy designed to reduce inflammation through a targeted molecular pathway. Difamilast belongs to a class of drugs known as phosphodiesterase-4 (PDE4) inhibitors, which regulate inflammatory mediators in the skin.
With the difamilast FDA approval, healthcare professionals now have an additional option to treat mild-to-moderate atopic dermatitis while minimizing steroid exposure. This advancement reflects the growing shift toward targeted dermatologic therapies that focus on immune modulation rather than broad immunosuppression.
In this comprehensive article, we will explore everything about difamilast FDA approval, including its mechanism of action, pharmacology, clinical trials, safety profile, side effects, drug interactions, and its role in modern dermatology.
Understanding Atopic Dermatitis
Atopic dermatitis (AD), commonly known as eczema, is a chronic inflammatory skin disease characterized by:
- Dry skin
- Severe itching
- Redness and swelling
- Skin thickening
- Recurrent flare-ups
The disease involves complex interactions between genetic predisposition, immune dysregulation, and environmental triggers.
Inflammatory cytokines such as IL-4, IL-13, and IL-31 play a key role in the pathophysiology of AD. Increased inflammatory signaling leads to persistent skin inflammation and itching.
Because of these mechanisms, modern therapies target specific inflammatory pathways, which is why the difamilast FDA approval represents a significant advancement.
What is Difamilast?
Difamilast is a topical phosphodiesterase-4 inhibitor developed for the treatment of mild-to-moderate atopic dermatitis.
It works by suppressing inflammatory signals in the skin and reducing cytokine production.
Key characteristics of difamilast include:
- Non-steroidal topical therapy
- PDE4 inhibitor
- Anti-inflammatory activity
- Suitable for long-term management
- Minimal systemic absorption
The difamilast FDA approval provides dermatologists with a targeted topical treatment that may offer improved safety compared to corticosteroids.
Difamilast Drug Class
Difamilast belongs to the phosphodiesterase-4 inhibitor (PDE4 inhibitor) class.
PDE4 enzymes regulate intracellular levels of cyclic adenosine monophosphate (cAMP). By inhibiting PDE4, difamilast increases cAMP levels inside immune cells, which reduces inflammatory cytokine production.
Other drugs in the PDE4 inhibitor category include therapies used for inflammatory diseases such as psoriasis and dermatitis.
The difamilast FDA approval expands the use of PDE4 inhibition in dermatologic treatments.
Mechanism of Action (MOA)
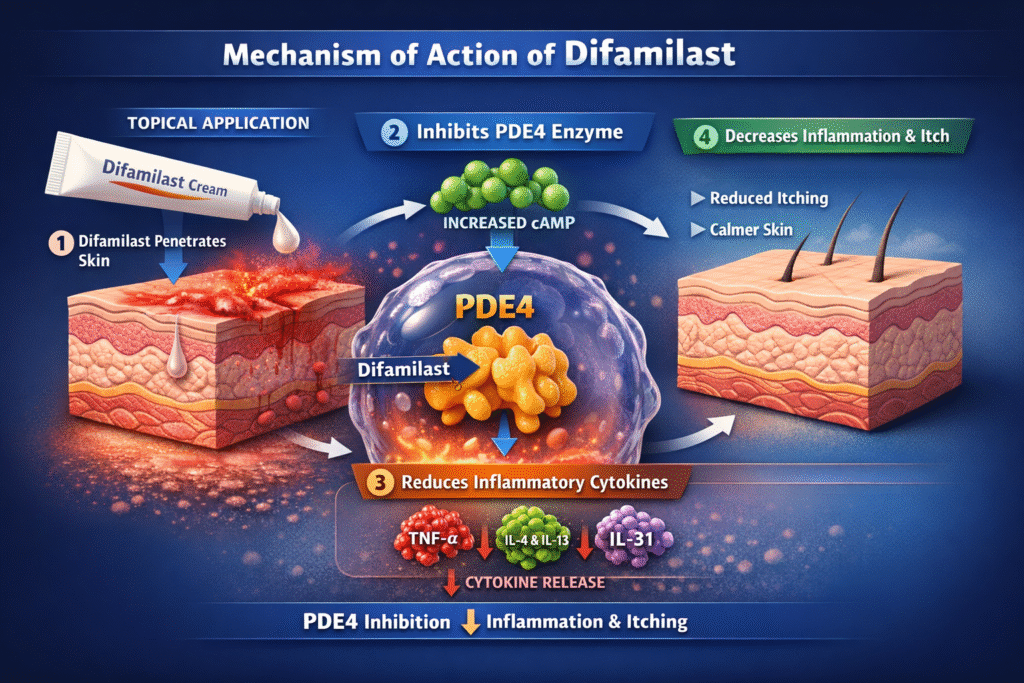
The mechanism of action of difamilast is centered on the inhibition of the PDE4 enzyme.
Step-by-Step Mechanism
- Difamilast penetrates the skin after topical application.
- The drug inhibits phosphodiesterase-4 (PDE4) enzymes inside inflammatory cells.
- PDE4 inhibition increases intracellular cAMP levels.
- Increased cAMP suppresses inflammatory cytokine production.
- Reduced cytokine release decreases skin inflammation and itching.
Cytokines Reduced by Difamilast
- Tumor Necrosis Factor (TNF-α)
- Interleukin-4
- Interleukin-13
- Interleukin-31
These cytokines are heavily involved in atopic dermatitis pathology.
Because of this targeted pathway, the difamilast FDA approval supports the use of precision anti-inflammatory therapy in dermatology.
Pharmacokinetics
Understanding the pharmacokinetics of difamilast is important for safe clinical use.
Absorption
After topical application, difamilast is absorbed into the skin layers where inflammation occurs.
Systemic exposure remains very low compared to oral medications.
Distribution
The drug primarily acts locally in the skin.
Minimal systemic distribution reduces the risk of systemic side effects.
Metabolism
Difamilast is metabolized mainly through enzymatic pathways in the body.
Elimination
Metabolites are excreted through normal metabolic processes.
The favorable pharmacokinetic profile further supports the clinical value of the difamilast FDA approval.
Clinical Trials Leading to Difamilast FDA Approval
The difamilast FDA approval was supported by multiple randomized clinical trials evaluating safety and efficacy in patients with atopic dermatitis.
Study Design
Clinical studies involved:
- Pediatric patients
- Adult patients
- Mild-to-moderate atopic dermatitis
- Randomized controlled trial design
Participants applied difamilast cream to affected areas over several weeks.
Key Clinical Outcomes
Important outcomes measured in the trials included:
- Reduction in eczema severity
- Improvement in itching
- Skin lesion improvement
- Safety evaluation
Patients receiving difamilast experienced significant improvement in eczema symptoms compared with placebo treatment.
These results were instrumental in obtaining difamilast FDA approval.
FDA Approval Details
The difamilast got FDA approval on 13 feb 2026 , represents a major milestone in dermatologic drug development.
- Approved for treatment of mild-to-moderate atopic dermatitis
- Topical cream formulation
- Suitable for both adults and pediatric patients
- Non-steroidal anti-inflammatory treatment
The difamilast FDA approval demonstrates the increasing demand for steroid-sparing therapies in dermatology.
Healthcare providers now have another option to treat eczema while minimizing long-term corticosteroid use.
Indications
According to the difamilast FDA approval, the medication is indicated for:
Treatment of mild-to-moderate atopic dermatitis in adults and children.
Patients experiencing eczema symptoms such as itching, inflammation, and skin lesions may benefit from difamilast therapy.
Dosage and Administration
Typical dosing recommendations include:
- Apply thin layer to affected skin
- Usually applied twice daily
- Avoid contact with eyes and mucous membranes
The dosing regimen established during the difamilast FDA approval process ensures effective symptom control with minimal adverse effects.
Side Effects
Like any medication, difamilast may cause side effects.
However, studies show that it has a favorable safety profile.
Common Side Effects
- Application site irritation
- Burning sensation
- Mild redness
- Dry skin
Less Common Side Effects
- Skin infection
- Allergic reactions
- Contact dermatitis
Because the medication is topical, systemic adverse effects are rare.
The safety profile observed during the difamilast FDA approval trials suggests it may be safer than long-term steroid therapy.
Drug Interactions
Drug interactions with difamilast are relatively limited due to its topical use.
However, clinicians should still consider possible interactions.
Potential Drug Interaction Considerations
- Topical corticosteroids – combined use may increase skin irritation
- Topical immunosuppressants – possible additive effects
- Other dermatologic medications – overlapping therapies should be monitored
During the evaluation process for difamilast FDA approval, no major systemic drug interactions were reported.
Advantages of Difamilast Over Steroids
The difamilast FDA approval offers several advantages compared to traditional steroid therapy.
Benefits
- Non-steroidal treatment
- Reduced risk of skin thinning
- Suitable for long-term use
- Targeted anti-inflammatory action
- Minimal systemic absorption
These benefits make difamilast an attractive option for chronic eczema management.
Role in Modern Dermatology
The difamilast FDA approval reflects the evolving landscape of dermatologic treatments.
In recent years, several targeted therapies have emerged, including:
- Biologic therapies
- JAK inhibitors
- PDE4 inhibitors
Difamilast adds to this growing group of precision dermatology treatments designed to control inflammation while minimizing systemic toxicity.
Future Research and Development
Following the difamilast FDA approval, researchers continue to explore additional applications for the drug.
Possible future research areas include:
- Use in pediatric eczema management
- Combination therapy with biologics
- Long-term safety monitoring
- Use in other inflammatory skin diseases
Ongoing studies may further expand the clinical role of difamilast.
Conclusion
The difamilast FDA approval represents a significant advancement in the treatment of atopic dermatitis. As a topical PDE4 inhibitor, difamilast offers a targeted anti-inflammatory approach that reduces eczema symptoms while minimizing the risks associated with long-term corticosteroid therapy.
Clinical trials supporting the difamilast FDA approval demonstrated meaningful improvements in skin inflammation, itching, and lesion severity in patients with mild-to-moderate atopic dermatitis. The drug also showed a favorable safety profile, with mostly mild and localized side effects.
For healthcare providers and dermatologists, the difamilast FDA approval provides an additional therapeutic option for managing eczema, particularly in patients who require steroid-sparing treatments.
As dermatology continues to move toward precision medicine, therapies like difamilast are expected to play an increasingly important role in improving patient outcomes and quality of life.

