Table of Contents
The Exdensur FDA approval represents a major advancement in the management of severe asthma, especially for patients suffering from the eosinophilic subtype of the disease. Severe asthma remains a challenging condition worldwide, often resistant to standard inhaled therapies. With the Exdensur FDA approval, patients and clinicians now have access to a highly innovative, ultra-long-acting biologic therapy that offers effective asthma control with only two doses per year.
This detailed guide explores everything you need to know about the Exdensur FDA approval, including how the drug works, who it is for, its clinical benefits, side effects, safety considerations, drug interactions, and its long-term significance in respiratory medicine.
Understanding Severe Eosinophilic Asthma
Asthma is a chronic inflammatory airway disease, but not all asthma is the same. In many patients with severe asthma, inflammation is driven by eosinophils, a type of white blood cell involved in immune responses. Elevated eosinophil levels contribute to:
- Persistent airway inflammation
- Increased mucus production
- Frequent asthma exacerbations
- Reduced lung function
- Poor response to conventional therapies
Despite high-dose inhaled corticosteroids and long-acting bronchodilators, many patients continue to experience uncontrolled symptoms. The Exdensur FDA approval directly addresses this unmet medical need by targeting the underlying inflammatory pathway rather than just managing symptoms.
What Is Exdensur?
Exdensur is a biologic monoclonal antibody therapy developed specifically for the add-on maintenance treatment of severe asthma with an eosinophilic phenotype. It is approved for use in adults and adolescents aged 12 years and older.
Unlike traditional asthma medications taken daily or weekly, Exdensur is designed for long-lasting activity, allowing it to be administered only twice a year. This feature alone makes the Exdensur FDA approval a landmark event in asthma treatment.
Key Characteristics of Exdensur
- Long-acting biologic therapy
- Targets immune-mediated inflammation
- Subcutaneous injection
- Administered by healthcare professionals
- Not intended for acute asthma attacks
Exdensur FDA Approval: Regulatory Overview
The Exdensur FDA approval was granted in December 2025 following a comprehensive review of clinical efficacy, safety, and manufacturing quality. The FDA approved Exdensur as an add-on maintenance therapy, meaning it is used alongside existing asthma treatments rather than replacing them.
Approved Indication
The Exdensur FDA approval covers:
- Severe asthma with an eosinophilic phenotype
- Patients aged 12 years and older
- Individuals inadequately controlled on standard asthma therapy
What the Approval Does NOT Cover
- Acute asthma exacerbations
- Emergency asthma treatment
- Rescue therapy
Patients must continue using short-acting bronchodilators for sudden symptoms.
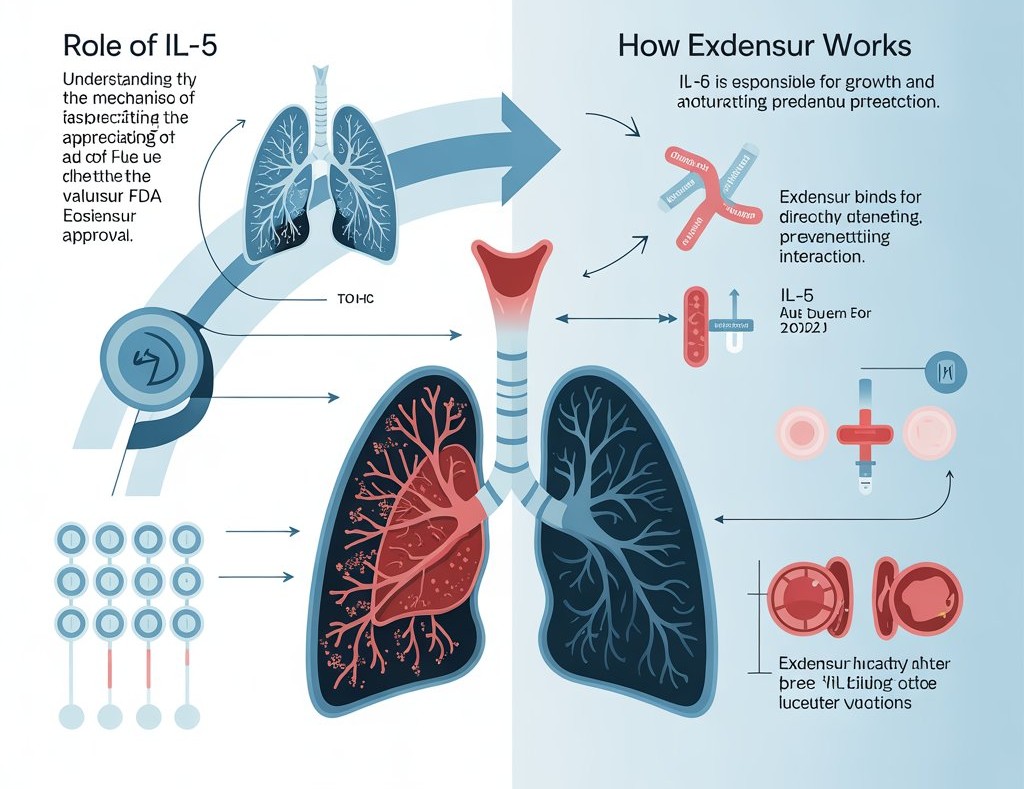
Mechanism of Action (MOA): How Exdensur Works
Understanding the mechanism of action is essential to appreciating the clinical value of the Exdensur FDA approval.
Role of Interleukin-5 (IL-5)
IL-5 is a cytokine responsible for:
- Growth and maturation of eosinophils
- Survival of eosinophils in blood and tissues
- Migration of eosinophils into lung airways
In severe eosinophilic asthma, IL-5 activity is abnormally high.
How Exdensur Targets IL-5
Exdensur is a monoclonal antibody that binds directly to IL-5, preventing it from interacting with its receptor. This leads to:
- Reduced eosinophil production
- Decreased eosinophil survival
- Lower airway inflammation
- Fewer asthma exacerbations
- Improved asthma control
The prolonged binding and stability of Exdensur explain why it remains effective for months, supporting the twice-yearly dosing schedule highlighted in the Exdensur FDA approval.
Clinical Trials Supporting Exdensur FDA Approval
The Exdensur FDA approval was based on robust evidence from large, well-designed phase III clinical trials involving patients with severe eosinophilic asthma.
Key Trial Outcomes
- Significant reduction in annual asthma exacerbation rates
- Decreased need for systemic corticosteroids
- Lower rates of emergency visits and hospitalizations
- Sustained asthma control over 12 months
- Consistent efficacy across age groups and severity levels
These outcomes confirmed that Exdensur not only reduces symptoms but also modifies disease activity at an immunological level.
Dosing and Administration
One of the most patient-friendly aspects of the Exdensur FDA approval is its dosing schedule.
Recommended Dosage
- Two subcutaneous injections per year
- Administered by a healthcare professional
- Typically given in a clinical setting
Advantages of Twice-Yearly Dosing
- Improved treatment adherence
- Reduced clinic visits
- Less treatment fatigue
- Greater convenience for patients and caregivers
This dosing frequency differentiates Exdensur from other biologics that require monthly or bi-monthly injections.
Side Effects of Exdensur
All medications carry some risk of side effects, and the Exdensur FDA approval includes clear safety guidance based on clinical trial data.
Common Side Effects
The most frequently reported side effects include:
- Upper respiratory tract infections
- Nasal congestion or allergic rhinitis
- Flu-like symptoms
- Sore throat
- Joint pain
- Injection-site reactions
These effects are generally mild to moderate and often resolve without treatment.
Serious Adverse Reactions and Warnings
Although uncommon, serious reactions can occur and are highlighted in the Exdensur FDA approval labeling.
Hypersensitivity Reactions
- Rash
- Swelling of face or throat
- Difficulty breathing
- Dizziness
Immediate medical attention is required if these symptoms appear.
Corticosteroid Tapering Warning
Patients using oral or inhaled corticosteroids should not stop them abruptly after starting Exdensur. Dose reduction must be gradual and supervised by a healthcare provider.
Parasitic Infections
Patients with active parasitic infections should receive treatment before initiating Exdensur, as eosinophils play a role in parasite defense.
Drug Interactions
One advantage highlighted by the Exdensur FDA approval is the drug’s low potential for significant drug-drug interactions.
Asthma Medications
Exdensur can be safely used with:
- Inhaled corticosteroids
- Long-acting beta-agonists
- Leukotriene modifiers
- Short-acting rescue inhalers
Other Medications
- No major interactions have been identified with common medications
- Caution is advised when used alongside strong immunosuppressive agents
- Patients should inform healthcare providers about all medications, supplements, and herbal products
Use in Special Populations
Pediatric Patients
The Exdensur FDA approval includes adolescents aged 12 years and older, providing a new option for young patients with severe asthma.
Pregnancy and Breastfeeding
- Limited human data available
- Use only if benefits outweigh potential risks
- Clinical decision-making should be individualized
Elderly Patients
No dose adjustment is required solely based on age.
Comparison With Other Asthma Biologics
The Exdensur FDA approval positions the drug uniquely among asthma biologics.
Key Differences
- Longest dosing interval among approved biologics
- Highly targeted IL-5 inhibition
- Reduced treatment burden
- Similar or superior efficacy in reducing exacerbations
For patients struggling with frequent injections or poor adherence, Exdensur may offer a significant advantage.
Impact of Exdensur FDA Approval on Asthma Care
The Exdensur FDA approval has broad implications for patients, clinicians, and healthcare systems.
Improved Quality of Life
- Fewer asthma attacks
- Reduced anxiety about symptom flare-ups
- Better sleep and daily functioning
Healthcare System Benefits
- Fewer emergency visits
- Lower hospitalization rates
- Reduced long-term steroid complications
Patient Selection: Who Benefits Most?
The ideal candidates following the Exdensur FDA approval include:
- Patients with documented eosinophilic asthma
- Individuals with frequent exacerbations
- Those uncontrolled on high-dose inhaled therapy
- Patients seeking less frequent dosing options
Proper diagnostic evaluation is essential before initiation.
Long-Term Safety and Monitoring
Patients receiving Exdensur should undergo:
- Regular asthma control assessments
- Monitoring for hypersensitivity reactions
- Periodic evaluation of eosinophil levels
- Review of overall medication regimen
Long-term studies continue to monitor safety outcomes post-approval.
Future Outlook After Exdensur FDA Approval
The Exdensur FDA approval signals a shift toward ultra-long-acting biologic therapies in chronic respiratory diseases. Ongoing research may expand its use to other eosinophil-driven conditions, further increasing its clinical relevance.
Conclusion
The Exdensur FDA approval marks a transformative moment in severe asthma management. By combining targeted immunotherapy with unprecedented dosing convenience, Exdensur addresses both clinical efficacy and real-world treatment challenges.
With its proven mechanism of action, favorable safety profile, minimal drug interactions, and twice-yearly dosing schedule, Exdensur offers hope to patients whose asthma remains uncontrolled despite standard therapy. As clinical experience grows, the Exdensur FDA approval is expected to reshape treatment strategies and significantly improve patient outcomes.

