Table of Contents
The nedosiran approval marks a significant milestone for patients suffering from Primary Hyperoxaluria Type 1 (PH1), a rare genetic disorder.
For individuals living with PH1, the accumulation of oxalate can lead to severe kidney damage and other systemic complications. The introduction of nedosiran approval as a treatment option brings new hope to these patients.

Nedosiran approval
This innovative treatment has shown promise in reducing oxalate production, thereby potentially mitigating some of the disease’s severe effects.
Key Takeaways
- nedosiran approval is a new treatment for Primary Hyperoxaluria Type 1.
- It aims to reduce oxalate production in patients.
- The nedosiran approval is a significant development for PH1 patients.
- Nedosiran may help mitigate severe kidney damage.
- It offers new hope for individuals living with this rare genetic disorder.
What is Nedosiran?
Nedosiran, a novel therapeutic agent, has been designed to target the root cause of Primary Hyperoxaluria Type 1 (PH1). It utilizes a cutting-edge approach to address this rare genetic disorder.
The Science Behind Nedosiran approval
Nedosiran approval is based on RNAi technology, a revolutionary method that allows for precise targeting of genetic material. This technology enables the drug to specifically inhibit the production of certain proteins involved in PH1.
Mechanism of Action
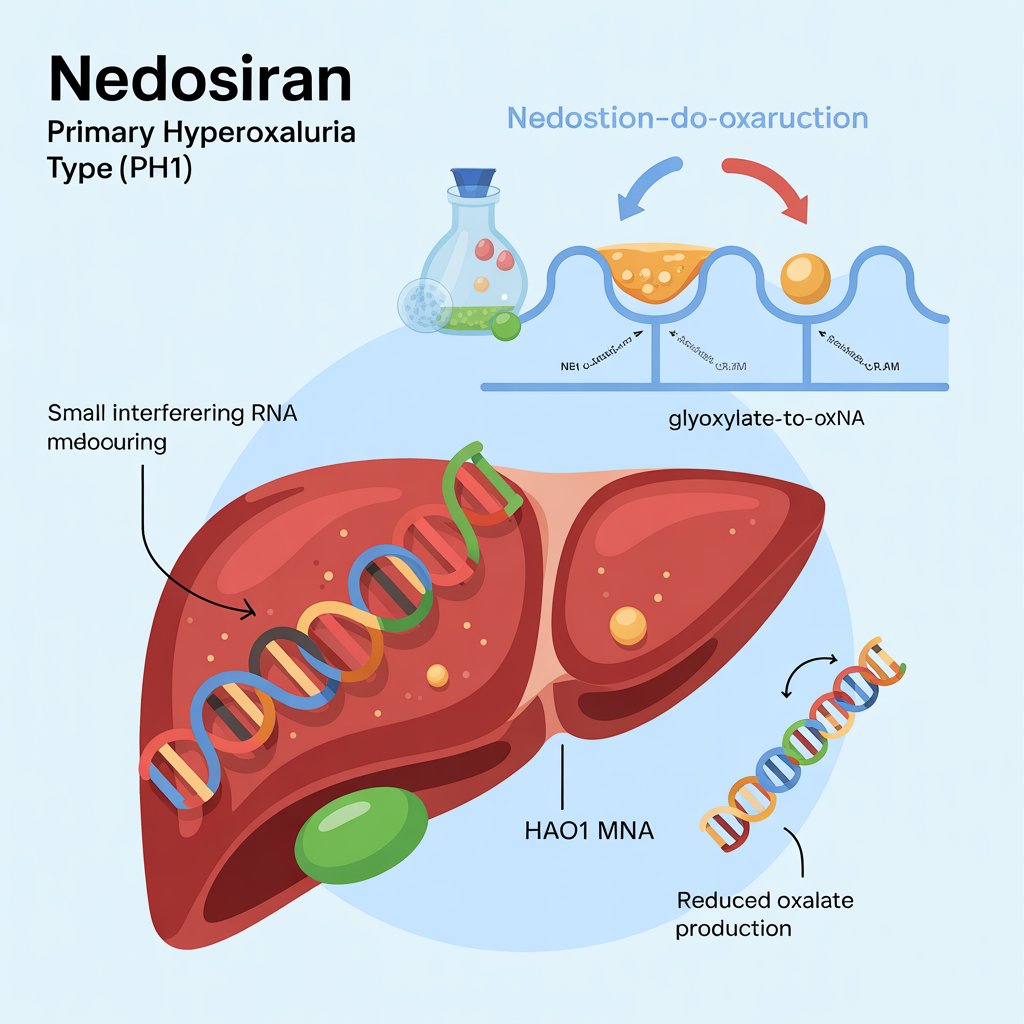
Nedosiran works by silencing specific genes responsible for the overproduction of oxalate, a hallmark of PH1. This is achieved through its RNA interference mechanism, which prevents the translation of target genes into proteins.
RNAi Technology Explained
RNAi technology is a biological process in which RNA molecules inhibit gene expression. Nedosiran leverages this natural process to reduce oxalate production, thereby addressing the underlying cause of PH1.
How Nedosiran Works in the Body
Once administered, nedosiran approval targets specific organs and systems involved in PH1. Its mechanism ensures that the drug has a lasting effect on reducing oxalate levels.
Target Organs and Systems
Nedosiran primarily targets the liver, where it inhibits the production of oxalate. This targeted approach minimizes potential side effects and maximizes efficacy.
Duration of Effect
The effect of Nedosiran can persist for an extended period, reducing the frequency of dosing required for effective management of PH1. This allows patients to maintain a more normal lifestyle.
Understanding Primary Hyperoxaluria Type 1 (PH1)
Understanding Primary Hyperoxaluria Type 1 requires a deep dive into its symptoms, progression, and treatment options. Primary Hyperoxaluria Type 1 (PH1) is a rare genetic disorder that significantly impacts the body’s ability to metabolize oxalate, leading to its accumulation.
Symptoms and Progression of PH1
The symptoms of PH1 can vary widely among patients, but they generally include signs related to kidney dysfunction. Early detection is crucial.
Early Warning Signs
Early warning signs of PH1 often involve recurrent kidney stones and nephrocalcinosis, a condition characterized by calcium deposits in the kidneys. These symptoms can manifest early in life.
Long-term Complications
If left untreated, PH1 can lead to severe longterm complications, including end stage renal disease and systemic oxalosis, where oxalate crystals deposit in various tissues throughout the body.
Current Treatment Landscape
The current treatment landscape for PH1 involves a combination of conservative management and, in some cases, more aggressive interventions.
Conventional Therapies
Conventional therapies include high fluid intake to help dilute the concentration of oxalate in the urine and potassium citrate to reduce the formation of kidney stones.
Limitations of Existing Treatments
Despite these measures, existing treatments have limitations, as they may not adequately address the underlying cause of oxalate overproduction. Thus, there’s a need for more targeted therapies.
Nedosiran Approval: Milestone for PH1 Treatment
Nedosiran approval marks a significant milestone in the treatment of Primary Hyperoxaluria Type 1 (PH1). This development brings new hope to patients suffering from this rare genetic disorder. nedosiran approval is backed by robust clinical trials that demonstrate its efficacy and safety.
Clinical Trials and Research Data
The PHYOX clinical trial was instrumental in assessing Nedosiran’s effectiveness. The trial results showed promising outcomes in reducing oxalate levels in patients with PH1.
PHYOX Clinical Trial Results
The PHYOX trial demonstrated that Nedosiran significantly reduced urinary oxalate levels, a key indicator of PH1 severity. This reduction is crucial for slowing disease progression.
Efficacy Metrics and Outcomes
Efficacy was measured through various metrics, including the reduction of urinary oxalate levels. The trial outcomes supported Nedosiran’s potential as a game-changer in PH1 treatment.
FDA Decision and Timeline
The FDA’s decision to nedosiran approval was influenced by the positive results from clinical trials. The Breakthrough Designation played a significant role in expediting the approval process.
Breakthrough Designation Process
Nedosiran approvalreceived the Breakthrough Therapy designation, which facilitated a faster review process. This designation is reserved for therapies that offer significant improvements over existing treatments.
Post-nedosiran approval Monitoring Requirements
Nedosiran approval will be subject to ongoing monitoring to ensure its continued safety and efficacy
This monitoring is crucial for maintaining patient safety and updating treatment guidelines as necessary.
Benefits of Nedosiran Treatment
Patients with PH1 can now benefit from Nedosiran, a treatment that has demonstrated efficacy in lowering oxalate levels and improving quality of life. This innovative therapy addresses a significant unmet need in the management of PH1.
Efficacy in Reducing Oxalate Levels
Nedosiran approval has shown remarkable efficacy in reducing oxalate levels in patients with PH1.Clinical trials have demonstrated a significant decrease in urinary oxalate excretion, a key marker of the disease.
Measurable Biomarker Improvements
The treatment has resulted in measurable improvements in biomarkers associated with PH1, providing a clear indication of its effectiveness. These improvements are crucial for monitoring disease progression and treatment response.
Timeframe for Expected Results
Patients can expect to see reductions in oxalate levels within a few weeks of starting Nedosiran treatment. The exact timeframe may vary depending on individual patient factors and the severity of their condition.
Quality of Life Improvements
Beyond reducing oxalate levels, Nedosiran has been associated with significant improvements in quality of life for patients with PH1. This includes reduced symptoms and a decreased need for dialysis.
Patient-Reported Outcomes
Studies have highlighted positive patient
reported outcomes, with many experiencing an enhanced overall wellbeing and reduced disease burden.
Reduced Need for Dialysis
By lowering oxalate levels, Nedosiran can help reduce the need for dialysis, a procedure that is often required for patients with advanced PH1. This reduction can significantly improve a patient’s quality of life.
| Benefit | Description | Clinical Impact |
| Reduced Oxalate Levels | Nedosiran decreases urinary oxalate excretion | Significant reduction in disease progression |
| Improved Biomarkers | Measurable improvements in PH1 biomarkers | Effective disease management |
| Enhanced Quality of Life | Patients experience reduced symptoms and improved well-being | Better overall health outcomes |
“The Nedosiran approval represents a major breakthrough in the treatment of Primary Hyperoxaluria Type 1, offering new hope to patients and their families.”
Potential Side Effects and Risks
Understanding the potential side effects of Nedosiran approval is crucial for patients considering this treatment. As with any medication, it’s essential to be aware of the possible risks and adverse reactions associated with its use.
Common Side Effects
Nedosiran may cause several common side effects, which can vary in severity. These include:
Injection Site Reactions
Some patients may experience reactions at the injection site, such as redness, swelling, or pain. These reactions are typically mild and resolve on their own.
Systemic Side Effects
Systemic side effects can include fatigue, headache, and nausea. These effects are generally manageable but should be reported to a healthcare provider if they persist or worsen.
Serious Adverse Reactions to Watch For
While less common, serious adverse reactions can occur. It’s crucial for patients to be aware of the warning signs that require medical attention.
Warning Signs Requiring Medical Attention
Symptoms such as severe abdominal pain, difficulty breathing, or signs of an allergic reaction (e.g., rash, itching) necessitate immediate medical evaluation.
Long-term Safety Considerations
Ongoing monitoring is necessary to understand the long-term safety profile of Nedosiran. Patients should follow up with their healthcare providers regularly to assess their condition and address any concerns.
| Side Effect | Frequency | Severity |
| Injection Site Reactions | Common | Mild |
| Fatigue | Common | Moderate |
| Severe Allergic Reactions | Rare | Severe |
Dosage and Administration Guidelines
Understanding the correct dosage and administration guidelines for Nedosiran is crucial for its effective use in treating Primary Hyperoxaluria Type 1 (PH1). The following information outlines the recommended dosing schedule and administration methods to ensure optimal therapeutic outcomes.
Recommended Dosing Schedule
The dosing schedule for Nedosiran approval is designed to achieve maximum efficacy in reducing oxalate levels. Patients are advised to follow the prescribed regimen closely.
Age-Based Dosing Considerations
Dosing for pediatric patients is adjusted based on age and weight, requiring careful calculation by healthcare providers to ensure safety and efficacy.
Dose Adjustments
Dose adjustments may be necessary based on patient response and tolerance. Regular monitoring by healthcare providers is essential to determine the need for any adjustments.
Administration Methods
Nedosiran can be administered via subcutaneous injection, a method that allows for flexibility in self-administration or administration by a healthcare provider.
Self-Administration Instructions
Patients or caregivers can be trained for self
administration following proper techniques and guidelines provided by healthcare professionals.
Healthcare Provider Administration
For those who prefer or require it, healthcare provider administration is available, ensuring that the treatment is administered correctly.
Dosage guidelines
Patient Eligibility for Nedosiran
Determining patient eligibility for Nedosiran is a crucial step in treating Primary Hyperoxaluria Type 1 (PH1) effectively. Nedosiran has been designed to target the root cause of PH1, and identifying suitable candidates is vital for maximizing its therapeutic benefits.
Who Should Consider This Treatment
Patients diagnosed with Primary Hyperoxaluria Type 1 should be evaluated for Nedosiran treatment. This includes individuals who have shown symptoms of PH1 or have a confirmed genetic diagnosis.
Genetic Testing Requirements
Genetic testing is a prerequisite for Nedosiran eligibility. Patients must have a confirmed diagnosis of PH1 through genetic testing that identifies mutations in the AGXT gene.
Disease Severity Considerations
The severity of PH1 can vary among patients. Healthcare providers assess disease severity to determine if Nedosiran is appropriate, considering factors such as oxalate levels and kidney function.
Contraindications and Precautions
While Nedosiran offers significant benefits, there are certain contraindications and precautions to be aware of. These include specific medical conditions and considerations for pregnant or breastfeeding women.
Medical Conditions Affecting Eligibility
Patients with certain medical conditions may not be eligible for Nedosiran. These conditions include severe kidney impairment or other significant health issues that could interact with the treatment.
Pregnancy and Breastfeeding Considerations
Pregnant or breastfeeding women should consult their healthcare provider about the potential risks and benefits of Nedosiran treatment. The safety of Nedosiran in these populations is still being studid.
Insurance Coverage and Cost Considerations
Insurance coverage for Nedosiran can be complex, but it’s a critical factor in accessing this treatmen. Patients need to understand their insurance options to manage costs effectively.
Navigating Insurance Approval
Navigating the insurance nedosiran approval process for Nedosiran involves several steps, including prior authorization and potential appeals. It’s essential for patients to work closely with their healthcare providers to ensure a smooth process.
Prior Authorization Requirements
Prior authorization is often required for Nedosiran approval treatment. This involves submitting documentation to the insurance provider to justify the medical necessity of the treatment. Patients should be prepared to provide detailed medical records and treatment plans.
Appeal Processes
If an insurance claim is denied, patients have the right to appeal. The Nedosiran approval process typically involves submitting additional information or clarifying the initial request. It’s crucial to follow the insurance provider’s appeal procedures carefully.
Patient Assistance Programs
For patients facing financial challenges, patient assistance programs can provide significant support. These programs can help reduce the out-of-pocket costs associated with Nedosiran treatment.
Manufacturer Support Programs
The manufacturer of Nedosiran may offer support programs to help patients access the treatment. These programs can include financial assistance, co-pay assistance, and other forms of support. Patients should inquire about these programs when discussing treatment options with their healthcare provider.
Non-Profit Resources
Nonprofit organizations dedicated to supporting patients with rare diseases like PH1 can also provide valuable resources. These organizations may offer grants, counseling, and other forms of assistance to help patients navigate the treatment process.
By understanding insurance coverage options and available patient assistance programs, patients can better manage the costs associated with Nedosiran treatment. It’s essential to explore all available resources to ensure access to this critical treatment.
Living with PH1: Lifestyle Recommendations
For individuals diagnosed with PH1, adopting the right lifestyle can significantly impact their quality of life. Managing this condition effectively requires a multifaceted approach that includes medical treatment, dietary adjustments, and regular monitoring.
Dietary Considerations
Diet plays a crucial role in managing PH1. Making informed dietary choices can help reduce oxalate levels and slow disease progression.
Fluid Intake Guidelines
Staying well-hydrated is essential for individuals with PH1. Aim for at least 8-10 glasses of fluid per day, and consider increasing intake during hot weather or when engaging in strenuous activities.
Foods to Limit or Avoid
Certain foods can exacerbate PH1 symptoms. It’s advisable to limit or avoid foods high in oxalate, such as spinach, beets, and rhubarb. Consulting a dietitian can help create a personalized meal plan.
Monitoring Your Condition
Regular monitoring is vital for managing PH1 effectively. This includes both home monitoring techniques and regular laboratory tests.
Home Monitoring Techniques
Individuals with PH1 can monitor their condition at home by tracking urine output and watching for signs of kidney stones or other complications.
Laboratory Test Schedule
Regular laboratory tests are crucial for assessing the effectiveness of the treatment plan and making necessary adjustments. Schedule regular check-ups with your healthcare provider to stay on top of your condition.
| Test Type | Frequency | Purpose |
| Urine Oxalate Levels | Every 3 months | To assess oxalate reduction |
| Kidney Function Tests | Every 6 months | To monitor kidney health |
| Imaging Studies | As needed | To check for kidney stones or damage |
What Patients Should Ask Their Healthcare Providers
To make informed decisions about their care, patients should inquire about several key aspects of Nedosiran approval treatment with their healthcare providers. This includes understanding the treatment’s expectations, potential side effects, and how it fits into their overall treatment plan.
Important Questions About Nedosiran
Patients should be prepared to ask about the specifics of Nedosiran treatment. This includes:
- How Nedosiran works to reduce oxalate levels
- The expected outcomes of the treatment
- Potential side effects and how to manage them
Treatment Expectations
Understanding what to expect from Nedosiran treatment is crucial. Patients should ask about the likelihood of achieving reduced oxalate levels and how this will impact their overall health.
Combination Therapy Options
It’s also important to inquire about whether Nedosiran can be used in conjunction with other treatments. Patients should ask about the potential benefits and risks of combination therapy.
Follow-up Care and Monitoring
Regular follow-up care is essential for patients undergoing Nedosiran treatment. This includes:
- Regular check-ups to monitor treatment efficacy
- Tests to assess oxalate levels and kidney function
Recommended Check-up Schedule
Patients should ask their healthcare providers about the recommended schedule for follow-up appointments and any necessary tests.
Signs of Treatment Success
Understanding the indicators of successful treatment will help patients monitor their progress and adjust their treatment plan as necessary.
Conclusion
Nedosiran approval is a significant breakthrough in the treatment of Primary Hyperoxaluria Type 1 (PH1). This innovative therapy has demonstrated its ability to reduce oxalate levels, offering new hope to patients suffering from this rare genetic disorder.
With Nedosiran approval, patients can expect a significant improvement in their quality of life. The treatment has been shown to be effective in clinical trials, and its approval by regulatory authorities is a testament to its safety and efficacy.
As the medical community welcomes Nedosiran as a new treatment option, it is essential for patients to discuss their eligibility and any questions they may have with their healthcare providers. By understanding the benefits and potential side effects of Nedosiran, patients can make informed decisions about their care.
The nedosiran approval is a result of rigorous research and clinical trials, and its impact on PH1 treatment will be significant. As patients and healthcare providers navigate this new treatment landscape, resources such as patient assistance programs and insurance coverage will play a crucial role in ensuring access to this life-changing therapy.
FAQ

