Table of Contents
Introduction
Cancer treatment has advanced dramatically in the past decades. Rather than relying on traditional chemotherapy – which indiscriminately kills rapidly dividing cells – new drugs target specific molecular pathways that tumor cells use to survive, grow, invade, and evade the immune system. One of the most promising innovations in this field is zanzalintinib, a next-generation oral tyrosine kinase inhibitor (TKI).
In this article, we will unravel the mechanism of action of zanzalintinib, explore how it interferes with tumor growth and spread, and understand its potential advantages over older treatments. This knowledge is especially valuable for pharmacy students, healthcare professionals, future prescribers, and anyone interested in modern cancer pharmacotherapy.
By reading this, you will gain a clear, clinically accurate, yet easy-to-understand view of how zanzalintinib works – from molecular interactions to its effects on cancer cells and the tumor microenvironment.
What is Zanzalintinib?
• Zanzalintinib (also known as XL-092) is an orally administered small-molecule TKI.
• It belongs to the class of multi-targeted receptor tyrosine kinase inhibitors.
• It is currently under investigation – in clinical development for a variety of advanced or metastatic solid tumors (such as kidney cancer, colorectal cancer, head and neck cancer, neuroendocrine tumors), either alone or in combination with immunotherapy.
• As a TKI, its therapeutic rationale lies in blocking several critical signaling pathways that tumors hijack for survival, growth, blood supply formation, metastasis, and immune evasion.
Thus, zanzalintinib is not just another chemotherapy – it is a precise, targeted therapy designed to disrupt cancer signaling and survival at the root.
Molecular Targets of Zanzalintinib
The hallmark of zanzalintinib is its ability to hit multiple critical receptor tyrosine kinases (RTKs) at once.
Key targets include:
- VEGFR2 (Vascular Endothelial Growth Factor Receptor 2)
- c-MET (also known as hepatocyte growth factor receptor, HGFR)
- TAM family kinases — especially AXL and MERTK (some data suggest other TAM kinases may be affected too)
- Possibly other related kinases (depending on tumor context)
These targets are deeply involved in processes like:
- Tumor angiogenesis (formation of new blood vessels)
- Tumor cell proliferation and survival
- Invasion, metastasis, and epithelial-to-mesenchymal transition (EMT)
- Crosstalk with the tumour microenvironment, including immune regulation and immune suppression
Because zanzalintinib simultaneously inhibits these RTKs, its mode of action is multi-pronged, addressing several hallmarks of cancer at once.
How Zanzalintinib Works: Mechanistic Pathways
Understanding the mechanism of action requires seeing how zanzalintinib interferes with different tumor-promoting processes.
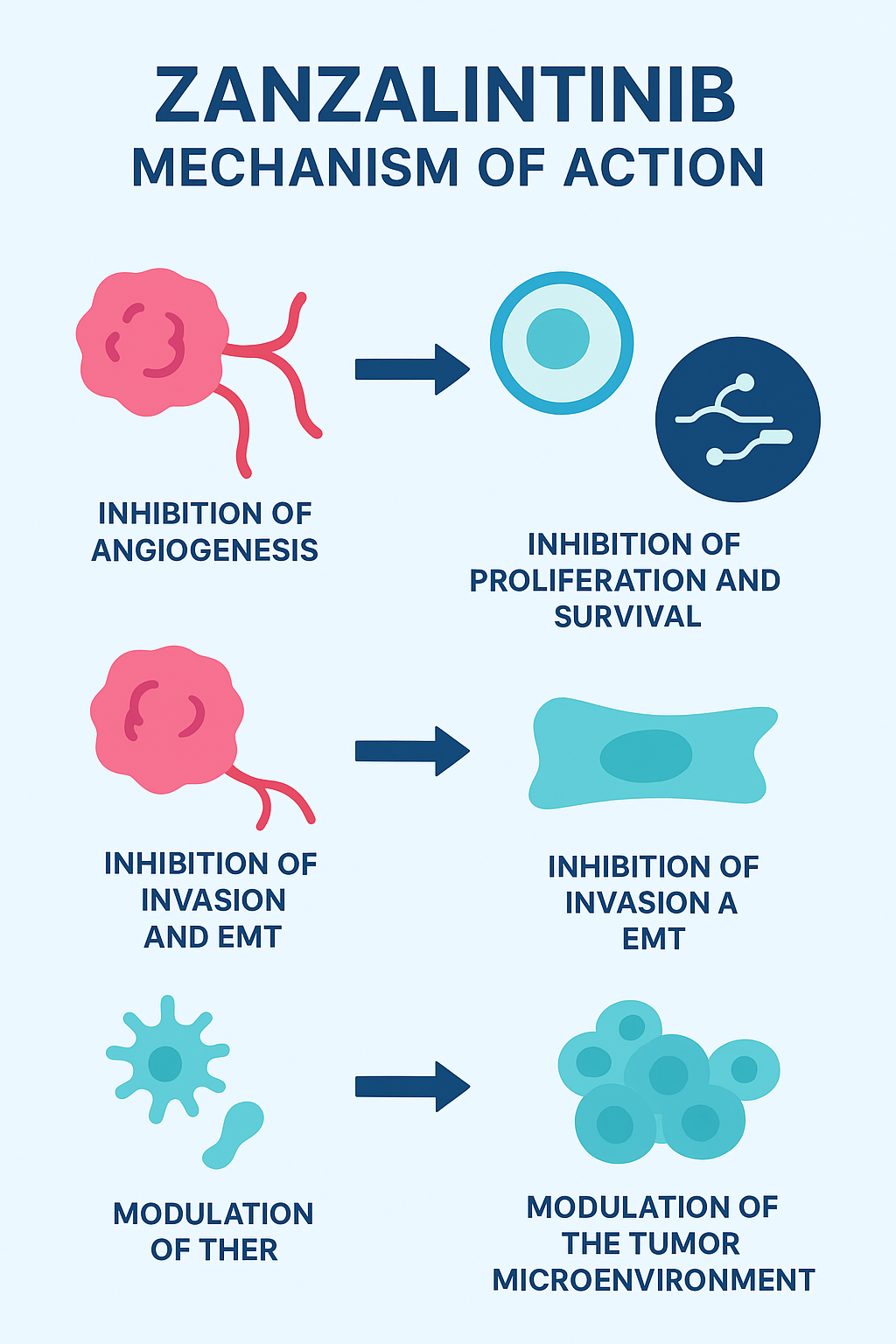
1. Inhibition of Angiogenesis (Cutting the Tumor’s Blood Supply)
• Tumors need a blood supply to grow — they secrete vascular growth factors (such as VEGF) that bind to receptors like VEGFR2 on endothelial cells. This initiates a cascade of events that lead to the formation of new blood vessels.
• Zanzalintinib binds to the kinase (ATP-binding) domain of VEGFR2, preventing receptor activation/phosphorylation. This blocks downstream signaling required for endothelial cell proliferation, migration, and new blood vessel formation.
• Result: Tumor vasculature is reduced, depriving tumors of oxygen and nutrients — limiting growth, survival, and potential metastasis.
• By disrupting angiogenesis, zanzalintinib weakens one of the fundamental requirements for tumor expansion.
2. Direct Anti-Tumor Effects: Inhibition of Proliferation, Survival, Invasion (via c-MET, AXL, MERTK)
• c-MET pathway: In many tumors, the ligand hepatocyte growth factor (HGF) binds to c-MET, which drives proliferation, survival, motility, invasion, and metastasis. Aberrant activation or overexpression of c-MET is a known driver in various cancers. By inhibiting c-MET, zanzalintinib disrupts this signaling, thereby inhibiting tumor growth and spread.
• AXL / MERTK (TAM kinases): These kinases contribute to aggressive tumor behavior, therapy resistance, EMT (epithelial-to-mesenchymal transition), invasion, and metastasis. High AXL/MERTK activity is often associated with poor prognosis, high metastatic potential, and resistance to therapy. Inhibition of these kinases by zanzalintinib may block EMT, reduce invasiveness, and potentially overcome resistance mechanisms.
• Since many tumor cells depend on one or more of these RTKs for survival and expansion, the broad targeting of zanzalintinib ensures more comprehensive blockade compared to single-target drugs.
3. Modulation of the Tumor Microenvironment & Immune Effects
One of the more exciting aspects of zanzalintinib is its potential to reshape the tumor microenvironment (TME) — not just by starving the tumor, but by making the environment less friendly for cancer, more friendly for immune attack.
• The RTKs targeted by zanzalintinib (particularly TAM kinases, MET, VEGFR) are not only localized to tumors or endothelial cells – they are often expressed on cells in the TME, including immune cells (macrophages, myeloid-derived suppressor cells, dendritic cells).
• Overactivation of these RTKs contributes to immunosuppression: by favoring regulatory T cells (Tregs), suppressive macrophages (M2 type), dendritic cell maturation or antigen presentation – helping tumors escape immune surveillance.
By inhibiting these RTKs, zanzalintinib can reduce immunosuppressive signaling, alter macrophage polarity, improve antigen presentation, and enhance lymphocyte (T cell) infiltration. In other words – the tumor can become “immune-permissive”.
• This reprogramming of the TME provides a strong rationale for combining zanzalintinib with immune checkpoint inhibitors (ICIs). When combined, the drug can weaken tumor defenses and help the immune system mount a more effective attack.
4. Preventing Resistance: Multi-Pathway Blockade
• Many older anti-angiogenic or single-pathway inhibitors face the problem of tumor adaptation or resistance. Tumors can switch to alternative signaling pathways (e.g., using the MET or TAM pathways when VEGF is inhibited), or undergo phenotypic changes (such as EMT) to survive.
• Because zanzalintinib simultaneously kills VEGFR2, MET, AXL, MERTK – changing the pathway makes it difficult for tumors to “escape”. This multi-target blockade reduces the likelihood of developing resistance, which can lead to more durable responses.
• In addition, broad-spectrum inhibition can simultaneously suppress metastasis, invasion, and immune escape mechanisms – addressing multiple hallmarks of cancer with a single drug.
Why Multi-Targeted TKIs Like Zanzalintinib Are a Paradigm Shift
To appreciate how significant zanzalintinib could be, we must compare it with older therapies and see what its multi-target strategy brings to the table.
Limitations of Older Single-Pathway Agents
- Many first-generation therapies focused solely on inhibiting angiogenesis (e.g., anti-VEGF agents) or a single receptor. While effective initially, resistance often developed because tumors used alternative pathways to survive, revascularize, or metastasize.
- Also, targeting a single pathway seldom addressed tumor cell invasiveness, immune evasion, or metastatic capabilities — limiting long-term efficacy.
- Moreover, tumors are not just a mass of malignant cells — they include supporting stromal cells, vasculature, immune cells, all contributing to growth and survival. Targeting only one aspect (like angiogenesis) does not sufficiently disrupt the complex tumor ecosystem.
Advantages of Zanzalintinib’s Multi-Pronged Approach
- Comprehensive blockade: By targeting multiple RTKs relevant to angiogenesis, proliferation, metastasis, and immune evasion, zanzalintinib addresses several cancer hallmarks at the same time.
- Reduced resistance potential: Simultaneous inhibition of alternate pathways makes it difficult for tumors to bypass the drug’s effects.
- Enhanced immunotherapy synergy: By remodeling the tumor microenvironment (reducing immunosuppression, improving vascular normalization, promoting immune infiltration), zanzalintinib sets the stage for more effective use of immune checkpoint inhibitors.
- Potential for durable responses: Multi-target inhibition may translate into longer disease control, better responses in aggressive or resistant cancers, and improved overall survival — especially in cancers with poor prognosis or limited treatment options.
In short: zanzalintinib epitomizes the shift from blunt-force chemotherapy to precision, multi-faceted cancer therapy.
Clinical Development and Context: Where Zanzalintinib Fits
Though not yet approved for routine clinical use, zanzalintinib is under intense investigation worldwide in multiple advanced solid tumors. Some clinical and development highlights:
- It is being studied as monotherapy and in combination with immune checkpoint inhibitors (ICIs) across diverse cancers — kidney cancer (particularly non-clear cell renal cell carcinoma, nccRCC), colorectal cancer, head & neck cancer, neuroendocrine tumours, and more.
- For kidney cancer (nccRCC), a phase III trial compares zanzalintinib plus an ICI versus standard-of-care therapy, aiming to show superiority in progression-free survival (PFS), objective response rate (ORR), and overall survival (OS).
- For colorectal cancer (especially refractory metastatic colorectal cancer), combination regimens (zanzalintinib + immunotherapy) show promise in improving survival and disease control compared to older therapies.
- Preclinical and early-phase clinical data suggest tolerable safety profiles, manageable toxicity, and encouraging antitumor activity. Importantly, compared with earlier multi-kinase inhibitors, zanzalintinib may offer improved tolerability because of optimized pharmacokinetics (e.g., possibly shorter half-life, greater kinase selectivity), which might reduce off-target effects.
Therefore, zanzalintinib stands at the forefront of a new wave of therapies aiming to deliver precision, efficacy, and improved safety, particularly for patients with limited options due to resistance or aggressive disease.
Strengths and Challenges of Zanzalintinib
Every novel therapy brings both promise and challenges. Understanding these helps both clinicians and pharmacists anticipate benefits and pitfalls.
Strengths
- Broad-spectrum targeting: Its ability to inhibit several critical RTKs makes it more versatile than narrow-spectrum drugs.
- Dual (or triple) mechanism: Anti-angiogenesis + direct tumor suppression + modulation of tumour microenvironment / immune effects.
- Synergy potential with immunotherapy: The TME-modulating effects create a fertile ground for combination therapy with ICIs.
- Potential for overcoming resistance and metastasis: By blocking pathways involved in invasion, EMT, metastasis, and immune escape, zanzalintinib may help in difficult-to-treat, therapy-resistant, or metastatic tumors.
- Improved tolerability (hypothetical): Compared to predecessors, optimized pharmacokinetic and pharmacodynamic properties may reduce dose-limiting toxicities and improve quality of life for patients.
Challenges and Considerations
- Investigational status: Zanzalintinib is not yet approved for standard clinical use. Until clinical trials complete and regulatory approvals are granted, its use remains limited to controlled settings.
- Long-term safety unknown: While early data suggest manageable toxicity, long-term side effects, rare adverse events, or cumulative toxicities remain to be fully characterized.
- Complexity of combination regimens: When combined with immunotherapy, the risk of overlapping toxicities, immune-related adverse events, and careful patient selection become critical.
- Potential drug resistance: Even with multi-target inhibition, tumors may eventually develop resistance (via mutations, bypass pathways, receptor upregulation, or downstream pathway activation). Understanding mechanisms of resistance will be key for long-term efficacy.
- Cost and access issues: As with many novel oncology agents — cost, availability, and access (especially in low- and middle-income countries) may limit widespread use.
Implications for Pharmacy Students, Healthcare Professionals & Future Pharmacists
Why should pharmacy students, pharmacists, and healthcare professionals care about zanzalintinib? Because this drug — and others like it — represent the future of oncology.
- Grasping the mechanism of multi-target TKIs: Understanding how RTKs work, and how their inhibition affects cancer’s multiple hallmarks, builds a solid pharmacology foundation.
- Preparing for patient counselling: When such drugs get approved, pharmacists will play a key role — explaining to patients how these differ from chemotherapy, what to expect, how to manage side effects, and the importance of compliance.
- Recognizing drug–drug interactions and monitoring needs: Multi-targeted TKIs can interact with other medications; also, because they can affect vasculature, liver function, immune system — regular monitoring will be essential.
- Supporting combination therapy: As oncology shifts toward combining targeted therapy + immunotherapy + supportive care, pharmacists will be critical in designing dosing schedules, managing side effects, and optimizing therapeutic outcomes.
- Contributing to research and pharmacovigilance: With new drugs come new learning opportunities. Pharmacists with knowledge of mechanisms can contribute to real-world evidence collection, adverse event reporting, and optimizing patient safety.
Thus, even in the academic phase, understanding zanzalintinib enriches your pharmaceutical knowledge, and prepares you for future roles in oncology pharmacy, clinical trials, patient education, and drug safety.
Conclusion
Zanzalintinib is a next-generation, multi-targeted oral tyrosine kinase inhibitor whose mechanism of action spans multiple critical dimensions of cancer biology: angiogenesis, tumor cell proliferation and survival, metastasis, immune evasion, and tumor microenvironment modulation.
By targeting VEGFR2, c-MET, AXL, MERTK (TAM kinases), and possibly related RTKs, zanzalintinib delivers a multi-pronged attack on cancer rather than a narrow strike on a single pathway. This broad-spectrum strategy offers multiple benefits — reducing tumor blood supply, reducing invasiveness, inhibiting metastatic spread, reactivating the immune system against cancer cells.
For pharmacy students, healthcare professionals, and future practitioners, understanding this mechanism is important. It not only represents a shift in how we treat cancer but also a paradigm where precision therapy + immunotherapy + supportive care together achieve better, more durable outcomes.
While zanzalintinib remains investigational, ongoing clinical trials offer hope — especially for patients with cancers that are resistant to standard treatments. As the evidence develops, this TKI could redefine treatment strategies for renal cell carcinoma, colorectal cancer, head and neck cancer, and beyond.

