Table of Contents
Introduction
The field of oncology has evolved dramatically in recent years, with the introduction of targeted therapies that offer improved outcomes with fewer side effects. Among these revolutionary therapeutic agents, pirtobrutinib stands out as a next-generation Bruton tyrosine kinase (BTK) inhibitor that offers new hope for patients battling advanced B-cell malignancies.
The pirtobrutinib approval has opened the door for patients with previously limited treatment options. Its development specifically targets challenges such as drug resistance and intolerance associated with previous BTK inhibitors.
What is Pirtobrutinib?
Pirtobrutinib (brand name: Jaypirca) is a highly selective, non-covalent (reversible) BTK inhibitor used in the treatment of hematological cancers.
Key Features
| Attribute | Description |
| Drug Type | Targeted therapy |
| Class | BTK inhibitor (Non-covalent) |
| Target | BTK protein |
| Resistance Profile | Effective against BTK C481 mutation |
| Route | Oral |
| Tolerance | High selectivity → reduced side effects |
This selectivity allows treatment continuation even after resistance develops to first-generation BTKi like Ibrutinib.
History and Timeline of Pirtobrutinib Approval
| Year | Key Milestone |
| Pre-2019 | Drug discovery and preclinical success |
| 2019 | Clinical trials begin (BRUIN trial) |
| Jan 2023 | Accelerated Pirtobrutinib approval by U.S. FDA |
| 2024–2025 | Expansion trials in multiple B-cell cancers |
The approval provided much-needed relief for patients with relapsed or refractory mantle cell lymphoma.
Importance of Pirtobrutinib Approval
Prior to the pirtobrutinib approval , patients who developed BTK inhibitor resistance had very limited options and were often faced with toxic chemotherapy.
Why the pirtobrutinib approval is important
✔ Works when most standard BTK inhibitors fail
✔ Oral dosing improves compliance and patient comfort
✔ Reduced toxicity compared to previous BTKIs
✔ Addresses a high unmet clinical need
✔ Long-term response durability demonstrates promise
This approval is considered a milestone in modern hematology.
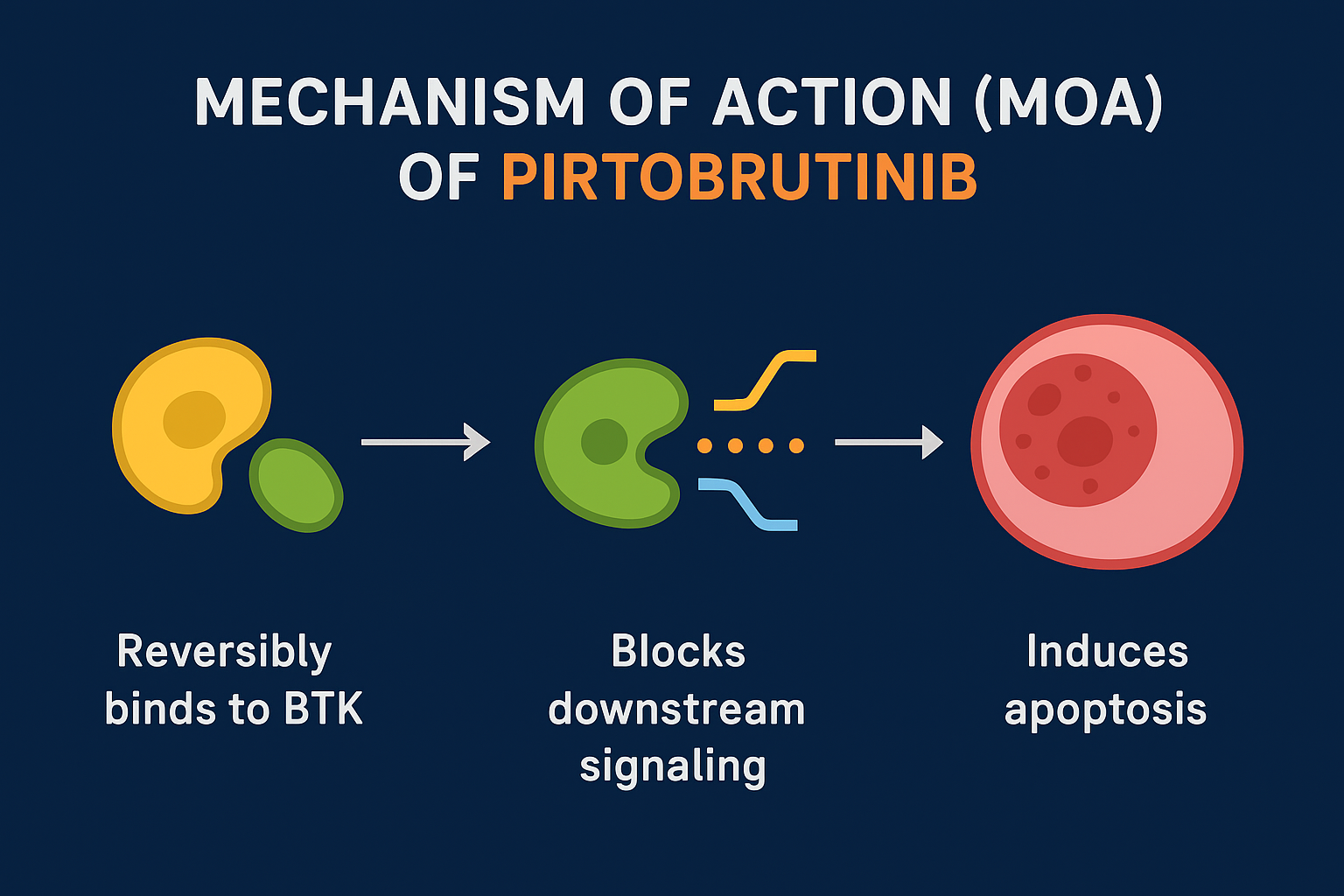
Mechanism of Action (MOA)
Pirtobrutinib blocks abnormal B-cell receptor signaling responsible for the growth and survival of cancer cells.
How it works
• Reversibly binds to BTK
• Active against both wild-type and mutated BTK
• Inhibits survival pathways including NF-κB, MAPK, PI3K
• Leads to apoptosis of malignant B-cells
✔ Reversible binding ensures activity even after the C481S mutation – a major cause of resistance with covalent BTKi.
Clinical Trial Data Supporting Approval
The FDA based Pirtobrutinib approval on findings from the global BRUIN Phase 1/2 trial.
Clinical Performance in Relapsed/Refractory Mantle Cell Lymphoma
| Outcome | Result |
| Overall Response Rate (ORR) | 50–60% |
| Complete Response (CR) | 13–20% |
| Duration of Response | >17 months |
| Median Progression-Free Survival (PFS) | ~7–9 months |
These outcomes are particularly impressive given that patients were heavily pretreated non-responders to other BTKi therapies.
FDA-Approved Indications
With Pirtobrutinib approval, the FDA allows its use in:
✔ Relapsed or Refractory Mantle Cell Lymphoma (MCL)
Patients must have received:
➡ At least two prior systemic therapies, including
➡ A BTK inhibitor
Additional approvals are expected as ongoing studies conclude.
Dosing and Administration
| Parameter | Recommendation |
| Dose | 200 mg once daily |
| Route | Oral tablets |
| Duration | Until progression or unacceptable toxicity |
| Food Restrictions | None significant |
The simple dosing schedule supports long-term patient adherence.
Side Effects and Management
Most patients tolerate treatment well, but some adverse reactions may occur.
Common Side Effects
- Fatigue
- Diarrhea
- Bruising or bleeding
- Cough
- Muscle and bone discomfort
- Cytopenias (low blood cell counts)
Serious Side Effects
Infections
Cardiac arrhythmias
Severe bleeding
Clinical monitoring is required:
- CBC at regular intervals
- Cardiac evaluation in high-risk cases
Early intervention helps maintain treatment continuity.
Drug Interactions
Pirtobrutinib is metabolized via CYP3A.
| Drug Type | Effect |
| Strong CYP3A inhibitors | ↑ Drug concentration → requires dose adjustment |
| CYP3A inducers | ↓ Effectiveness → avoid |
Patients should always inform physicians about herbal products and OTC drugs.
Use in Special Populations
| Group | Safety Consideration |
| Elderly | Generally well tolerated |
| Renal impairment | No major dose modifications needed |
| Hepatic impairment | Use with caution |
| Pregnancy | Avoid — fetal risk possible |
| Breastfeeding | Not recommended |
Comparison with Other BTK Inhibitors
| Feature | Earlier BTK inhibitors | Pirtobrutinib |
| Binding | Irreversible | Reversible |
| Effect on C481 mutation | Poor | Excellent |
| Off-target toxicity | Higher | Lower |
| Use after BTKi failure | Limited | Strong |
✨ This comparison highlights a major reason behind clinical excitement after Pirtobrutinib approval.
Access and Health Economics
Access varies by geography:
| Region | Status |
| United States | Available via insurance & assistance programs |
| Europe | Gradual rollout |
| India & Asian countries | Under regulatory evaluation |
| LMICs | Cost remains a major challenge |
Programs are emerging to help patients receive treatment early.
Research Beyond Mantle Cell Lymphoma
Ongoing clinical studies include:
- CLL / SLL
- Waldenström Macroglobulinemia
- Follicular Lymphoma
- Richter’s Transformation
- Combination with Venetoclax and monoclonal antibodies
More expanded indications are expected in the near future.
Real-World Outcomes
After Pirtobrutinib approval, physicians note:
✔ Faster symptom control (night sweats, fatigue)
✔ Better disease stability
✔ Improved quality of life
✔ Fewer hospitalizations
Patients often share restored energy levels and improvement in daily functioning
Challenges After Approval
Although a breakthrough, challenges persist:
- Drug cost remains high
- Long-term resistance may still appear
- Wider access needed across developing nations
- Combination therapy costs can be high
Continued research and pricing adjustments are expected to address these issues.
Future Outlook
Experts believe Pirtobrutinib will:
✨ Move into earlier lines of therapy
✨ Earn approvals across multiple B-cell cancers
✨ Become part of standard combination regimens
✨ Improve survival outcomes globally
It is set to reshape oncology guidelines and patient care strategies.
Conclusion
The pirtobrutinib approval is a major advance in precision oncology. It addresses a critical unmet need for patients with resistant B-cell malignancies, particularly mantle cell lymphoma. With compelling clinical performance, favorable safety profile, and expanded research prospects, pirtobrutinib stands as a promising next-generation treatment for hematologic cancer patients worldwide.
Key Points
✔ Breakthrough therapy for relapsed MCL
✔ Offers hope after other BTKI therapies have failed
✔ Superior design = better targeting + fewer side effects
✔ Positioned for expansion across multiple cancer types
✔ Represents a major leap in personalized cancer care

