Table of Contents
What is Lurbinectedin?
Lurbinectedin is an innovative anticancer drug primarily used to treat metastatic small cell lung cancer (SCLC) in adults who have received platinum-based chemotherapy. SCLC is known to be the most aggressive and rapidly spreading lung cancer, with a high recurrence rate and very few effective second-line treatments.
Due to this therapeutic gap, the approval of Lurbinectedin brought new hope to patients and oncologists. It is available under the brand name Zepzelka® and is administered as an intravenous infusion in a hospital setting by oncology specialists.
A major advantage of Lurbinectedin is its dual action – it not only directly targets cancer cells but also affects the tumor microenvironment, which provides better control of tumor growth.
FDA Approval :
In June 2020, Lurbinectedin received accelerated FDA approval as a second-line option in metastatic SCLC. The approval was based on strong clinical evidence of tumor shrinkage and symptom relief in patients who relapsed after first-line therapy.
Confirmatory studies are ongoing to collect long-term survival results by 2025. If successful, Lurbinectedin could receive:
• Full FDA approval status
• Expanded indication in prior-line therapy
• Broader use in combination regimens
The approval underscores how important Zepzelka has become in the modern SCLC treatment landscape, where treatment options are extremely limited.
Mechanism of Action
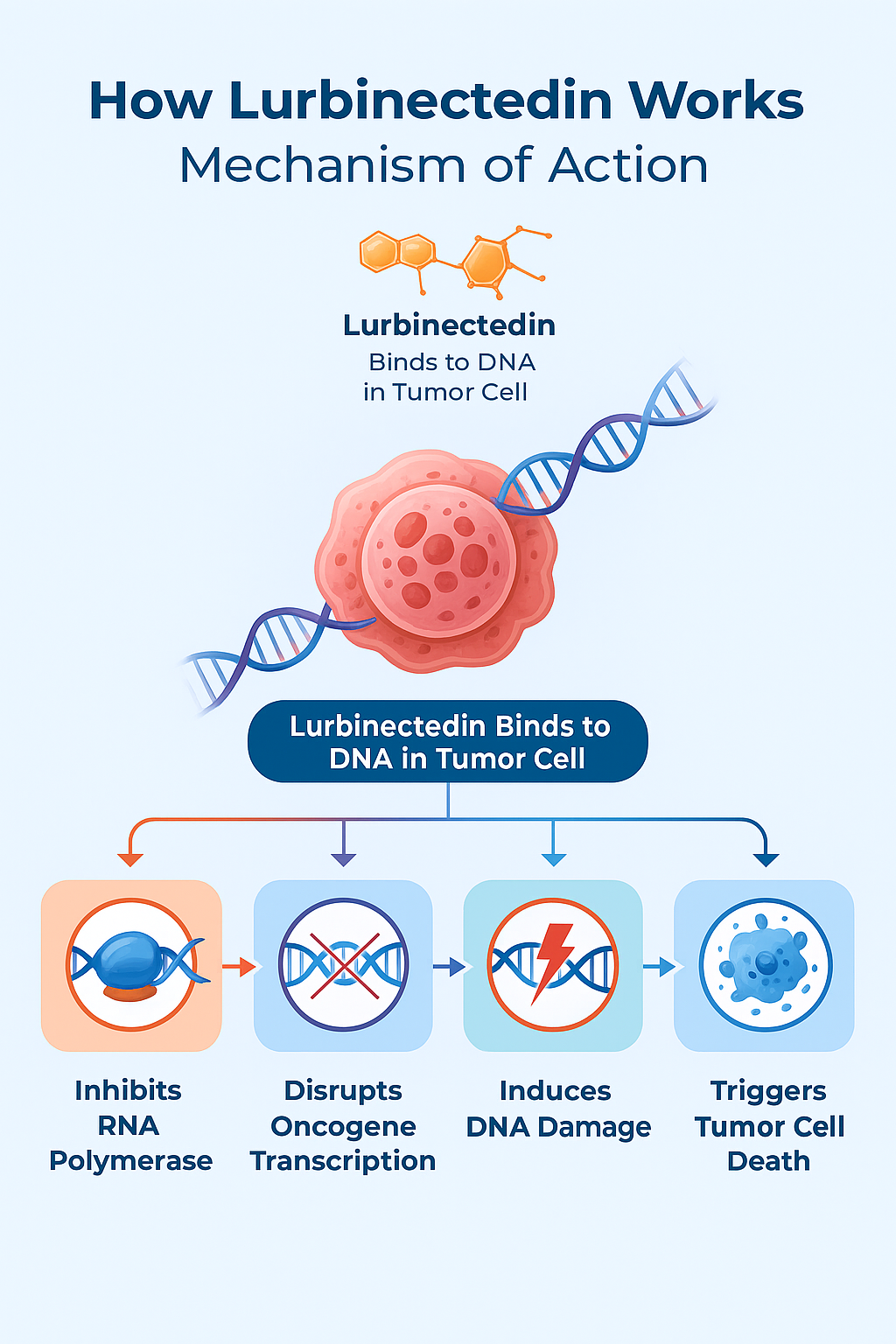
Zepzelka is classified as a selective inhibitor of oncogenic transcription.
Its mechanism of action includes the following key actions:
🔹 Directly binds to the DNA minor groove
This prevents cancer cells from transcribing essential growth-related genes.
🔹 Inhibits RNA polymerase II
This shuts down growth signals and protein production in tumor cells.
🔹 Induces DNA damage and apoptosis
Cancer cells are forced into programmed cell death, which stops tumor growth.
🔹 Reduces tumor-associated macrophages
These immune cells often help cancer grow — Lurbinectedin reduces them to weaken the cancer environment.
This combination makes Lurbinectedin different from conventional chemotherapy, which often targets cell division but not transcription.
Usage
Zepzelka is used to treat:
✔ Adults with metastatic SCLC
✔ Who have progressed on or after platinum-based therapy
✔ Including those who previously received immunotherapy
It is often chosen when:
- The disease returns aggressively after initial treatment
- Other chemotherapy drugs cause excessive toxicity
- Treatment options are limited due to poor patient tolerance
Doctors evaluate overall health, blood values, and organ function before starting therapy.
Dosage & Administration
- Dose: 3.2 mg/m²
- Route: Intravenous infusion into a vein
- Time: Over 60 minutes
- Schedule: Once every 21 days (3-week cycle)
Patients may need:
- Anti-nausea medication before infusion
- Dose modifications if severe fatigue or low blood counts develop
Doctors may temporarily delay a treatment cycle if needed to protect patient safety — a standard practice with many chemotherapy agents.
Clinical Trial Outcomes
Clinical studies have shown promising results for patients with relapsed SCLC:
Overall Response Rate: Around 35%
Median Duration of Response: About 5 months
Other key findings:
- Helps control liver and brain metastases in some patients
- Improves cancer-related symptoms like breathlessness and chest pain
- Shows better tolerability than older drugs like topotecan
For many patients, Lurbinectedin becomes a crucial lifeline after first-line therapy fails.
Side Effects and Safety
Like most cancer treatments, Lurbinectedin may cause side effects. Monitoring and supportive care help manage them effectively.
Common side effects
- Neutropenia — increases infection risk
- Fatigue and weakness
- Nausea and vomiting
- Hair thinning
- Constipation
- Loss of appetite
- Elevated liver enzymes
Serious but less common side effects
- Febrile neutropenia (fever with infection)
- Severe liver problems
- Rare lung inflammation (requires urgent care)
Immediate medical attention is required if the patient develops fever, chills, or unexplained lung symptoms.
Drug Interactions and Warnings
Lurbinectedin is broken down by CYP3A4, so interactions are important.
Avoid medicines like:
- Ketoconazole, clarithromycin → can increase side effects
- Rifampin, St. John’s Wort → can reduce effectiveness
Additional precautions:
- Avoid alcohol, which can worsen liver toxicity
- Avoid live vaccines during therapy
- Contraception must be used — drug can harm a fetus
Doctors evaluate medications carefully before prescribing Lurbinectedin.
Who Should Not Take Lurbinectedin?
Lurbinectedin may not be recommended in:
| Group | Why |
| Pregnant patients | Risk of fetal harm |
| Breastfeeding mothers | Drug may pass into milk |
| Severe hepatic impairment | Higher risk of toxicity |
| Weak immune system cases | Higher infection risk |
Elderly patients may receive Lurbinectedin, but require closer monitoring.
Monitoring During Treatment
To ensure safe use, clinicians regularly check:
- CBC (neutrophils, hemoglobin, platelets)
- Liver and kidney function
- Symptoms of infection or bleeding
- Tumor response through scans
- Overall physical condition and appetite
Monitoring schedules vary based on tolerance and treatment response.
Cost, Accessibility & Patient Support (US Focus)
As a specialty oncology drug, Lurbinectedin can be expensive.
However, multiple support programs help reduce the financial burden:
- Co-pay savings for insured patients
- Foundation support for low-income patients
- Financial assistance for uninsured cases
Hospital social workers or oncology support teams help patients apply.
Future Research and 2025 Pipeline Updates
Researchers are studying whether Lurbinectedin can provide even better outcomes when combined with other modern therapies. Current studies include:
- Lurbinectedin + immunotherapy
(e.g., pembrolizumab, atezolizumab) - First-line treatment trials in newly diagnosed SCLC
- Testing in other cancers, such as:
- Ovarian cancers
- Mesothelioma
- Breast cancer
- Neuroendocrine tumors
If results remain positive, Lurbinectedin may soon play a much larger role in cancer therapy.
Patient Counseling Advice
Patients should receive clear guidance on:
✔ Reporting fever or infection symptoms immediately
✔ Avoiding crowded spaces during low-immunity periods
✔ Staying hydrated + maintaining nutrition
✔ Planning rest because fatigue is common
✔ Careful contraception during and after therapy
✔ Attending every follow-up and lab appointment
Good communication improves outcomes significantly.
Final Conclusion
Lurbinectedin has emerged as an important option for patients with metastatic small cell lung cancer, especially when the disease recurs early after initial treatment. Its ability to target cancer cells and their surrounding environment makes it a powerful addition to the limited arsenal available for SCLC.
As research continues into 2025 and beyond, Lurbinectedin could open up new treatment avenues, offer early treatment options, and potentially expand to other difficult-to-treat cancers. For patients and families battling SCLC, this represents meaningful progress and much-needed hope.

