Table of Contents
The year 2025 marked a significant milestone in cancer treatment, redefining how physicians approach immune-based therapies. With the approval of several Next Gen Immunotherapy Approved in 2025, the oncology landscape shifted toward more precise, accessible, and patient-friendly interventions.
These innovations go beyond first-generation immunotherapies (such as classic checkpoint inhibitors) and introduce improved cell therapies, bispecific antibodies, ADCs, and subcutaneous checkpoint inhibitors, making treatment faster and more effective. This comprehensive guide analyzes each advancement, its clinical relevance, and how these next-generation therapies impact patients and physicians.
What Is Meant by “Next Gen Immunotherapy”?
“Next-generation immunotherapy” refers to a modern category of immune-based cancer treatments designed to overcome the limitations of older immunotherapies. These therapies are:
1. More Targeted
They selectively attack tumor cells with less harm to healthy tissues.
2. More Controlled
Engineered components reduce toxicity such as CRS or neurotoxicity.
3. More Convenient
New delivery routes—like subcutaneous checkpoint inhibitors—reduce infusion times.
4. More Accessible
Off-the-shelf immunotherapies such as bispecific antibodies eliminate the need for personalized cell engineering.
These advances explain why the phrase next gen immunotherapy approved in 2025 has become a defining highlight in oncology news and journals.
Major FDA Approvals: Next Gen Immunotherapy Approved in 2025
Below are the most influential advancements and approvals that shaped 2025.
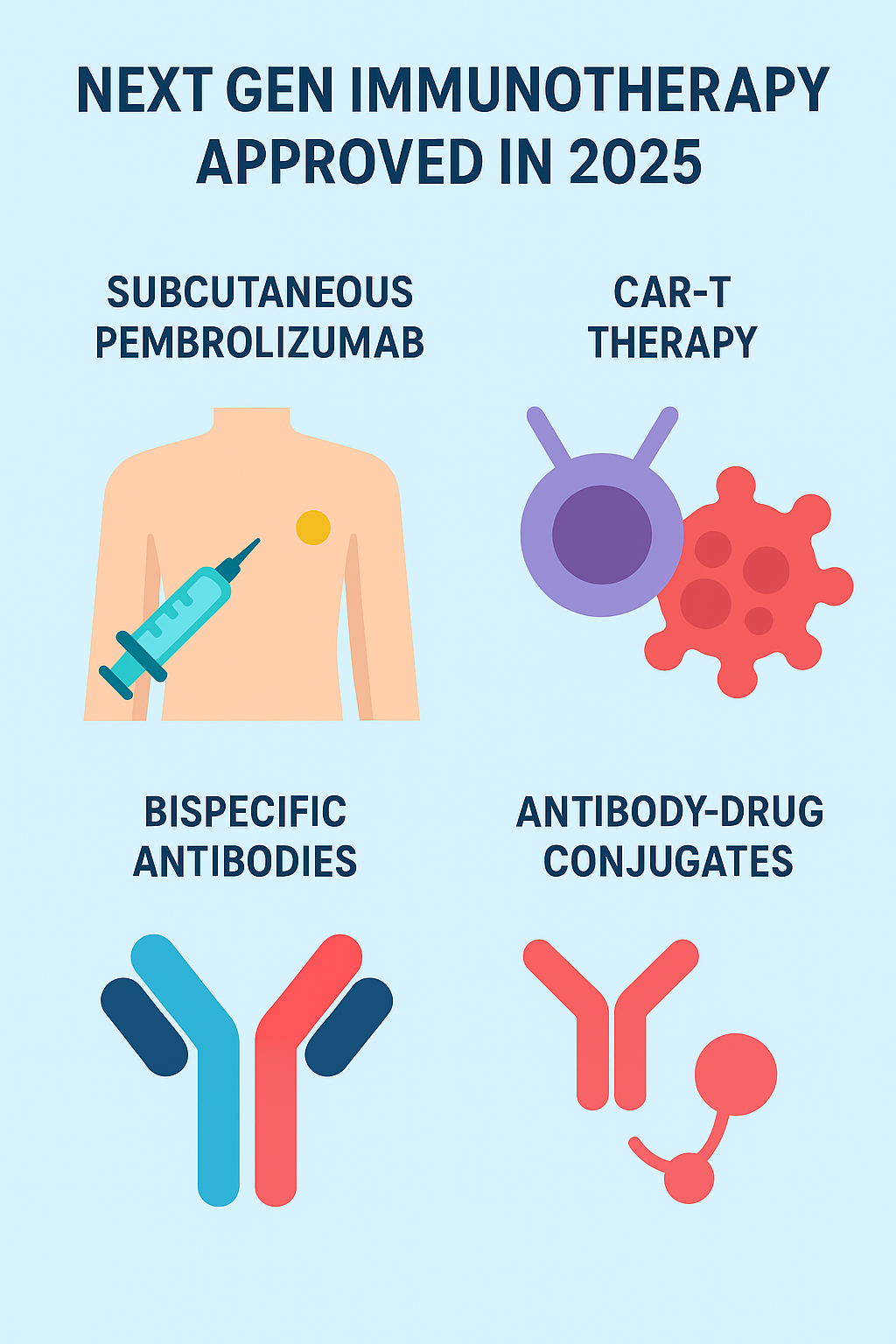
1. Subcutaneous Pembrolizumab (Keytruda Qlex) – A Game-Changing Approval
One of the most talked-about next gen immunotherapy approved in 2025 was the FDA approval of Keytruda Qlex, a subcutaneous formulation of pembrolizumab.
Why this approval matters:
- Replaces long IV infusions with quick under-the-skin injections
- Reduces time patients spend in infusion chairs
- Expands accessibility in community oncology centers
- Ideal for patients with limited venous access or infusion anxiety
This formulation does not change the drug’s mechanism but modernizes how patients receive immunotherapy, representing a major leap for convenience and quality of life.
2. CAR-T Therapy Label Expansions and Streamlined REMS Requirements
CAR-T therapy continued its rapid evolution in 2025, with multiple approvals and updates.
Key changes in 2025:
- Expanded indications for hematologic malignancies
- Earlier-line use in specific cancers
- Updated REMS programs showing improved safety profiles
- More predictable management of CRS and ICANS
- Consideration of outpatient administration in select centers
CAR-T therapies remain powerful, especially for patients who have failed multiple lines of treatment. The 2025 regulatory updates indicate increasing confidence in their safety and practicality.
This category contributes significantly to the list of next gen immunotherapy approved in 2025.
3. Rise of Bispecific Antibodies (BsAbs / T-Cell Engagers)
Bispecific T-cell engagers (TCEs) have gained tremendous traction. In 2025, several bsAbs received:
- Accelerated approvals
- Fast-track designations
- Expanded indications
These molecules simultaneously bind a cancer cell and a T cell, forcing immune engagement without the need for engineered cells.
Advantages of next-gen bispecifics:
- Off-the-shelf availability
- Stronger T-cell activation
- More manageable toxicity
- Potential to replace CAR-T in certain settings
In 2025, bispecifics were increasingly used in lymphomas, leukemias, and early solid tumor indications.
4. New ADCs (Antibody-Drug Conjugates) & Multispecific Antibodies
Antibody-Drug Conjugates continued their rise with multiple 2025 milestones.
Improvements seen in next-gen ADCs:
- Highly stable linkers
- Precision cytotoxic payloads
- Lower off-target toxicity
- Applications in GI cancers, breast cancer, and gynecologic cancers
Multispecific antibodies also received approvals and regulatory designations, enabling:
- Dual or triple targeting
- Enhanced immune synapse formation
- Improved response rates in resistant tumors
The next gen immunotherapy approved in 2025 expanded treatment options in several aggressive cancers.
Why 2025 Was a Landmark Year for Immuno-Oncology
The surge in next gen immunotherapy approved in 2025 was not random—it was the result of several scientific and regulatory trends:
1. Improved Manufacturing
Cell therapy manufacturing became faster and more reliable.
2. Better Safety Management
With strong CRS-management protocols, clinicians can treat patients more confidently.
3. Demand for Convenient Treatment Options
Patients increasingly prefer faster, injection-based therapies over hours-long infusions.
4. Stronger Biomarker-Based Approaches
More approvals came with companion diagnostics.
5. Increased Use of Combination Therapy
2025 saw more combinations involving:
- Checkpoint inhibitors
- Tumor vaccines
- Radiation
- ADCs
- Targeted therapies
Together, these factors accelerated the rollout of next-gen immunotherapies.
Impact on Clinical Practice
Here’s how the next gen immunotherapy approved in 2025 is transforming real-world oncology practice:
1. Faster Treatment Administration
Subcutaneous checkpoint inhibitors reduce chair time from hours to minutes.
2. More Treatment Options for Refractory Patients
Next-gen CAR-T and bispecifics provide hope for patients with previously incurable cancers.
3. Changing Toxicity Management
CRS protocols have become standardized and effective.
4. Wider Access to Cutting-Edge Therapies
Off-the-shelf TCEs and ADCs are now available even in mid-tier oncology centers.
5. Earlier Use of Immunotherapy
Some new approvals moved immunotherapy from late-stage salvage therapy into earlier treatment lines.
Patient Perspective: Benefits and Considerations
From a patient viewpoint, the next gen immunotherapy approved in 2025 offers several advantages:
Benefits:
- Shorter treatment times
- Improved tolerability
- Fewer hospital visits
- More personalized therapy
- Better outcomes, especially in blood cancers
Challenges:
- High cost of CAR-T and ADCs
- Immune toxicity remains a concern
- Limited access in certain geographic areas
- Need for biomarker testing to identify eligible patients
Overall, patients benefit from more tailored and convenient options.
Future Trends in Next-Gen Immunotherapy Beyond 2025
The 2025 breakthroughs are only the beginning. Several major trends are expected to continue shaping oncology:
1. Solid Tumor CAR-T Therapies
Technologies targeting TME suppression and antigen escape will expand CAR-T use beyond blood cancers.
2. Multi-Specific Antibodies
Tri-specific antibodies will outperform classic bispecifics.
3. AI-Powered Immunotherapy Development
AI will enhance:
- Neoantigen prediction
- Response modeling
- Personalized treatment strategies
4. Next-Generation ADC Payloads
Ultra-precise cytotoxic payloads with reduced systemic exposure are advancing rapidly.
5. Injectable & Oral Immunotherapies
More checkpoint inhibitors may switch to subcutaneous or oral forms.
The momentum of next gen immunotherapy approved in 2025 is expected to trigger further approvals in 2026 and 2027.
Limitations of Current Next-Gen Immunotherapies
Even though 2025 was exceptional, challenges remain:
- Long-term survival data is still maturing
- Some approvals were accelerated and require confirmatory trials
- High treatment costs limit access
- Infrastructure demands for CAR-T remain high
Recognizing these challenges is essential for realistic expectations and responsible clinical use.
Conclusion
The phrase next gen immunotherapy approved in 2025 marks a significant milestone in the evolution of cancer treatment. From subcutaneous checkpoint inhibitors to cutting-edge CAR-T advances, 2025 saw breakthroughs that improved patient access, increased convenience, and provided better outcomes for difficult-to-treat cancers. With improved delivery systems, smarter biologics, and a growing portfolio of multispecific antibodies and ADCs, the future of immunotherapy looks more promising than ever. Healthcare professionals, cancer patients, and researchers will all benefit from these transformative innovations.