Table of Contents
Introduction: Why “Nitisinone FDA Approval” Is Making Headlines
The topic of nitisinone FDA approval has been hotly debated following the recent decision by the US Food and Drug Administration (FDA) to approve Harliku (nitisinone) for the treatment of a rare metabolic disorder – alkaptonuria (AKU) – in 2025.
Originally introduced in 2002 under the brand name Orfadin for hereditary tyrosinemia type 1 (HT-1), nitisinone has evolved into a game-changing therapy for rare metabolic conditions. Its approval for a new indication is a significant milestone in the history of orphan drug development.
Let’s explore the full story – from the mechanism of action and regulatory journey to clinical trials, safety and the real significance of this groundbreaking drug.
What Is Nitisinone?
Nitisinone is a small-molecule inhibitor of the enzyme 4-hydroxyphenylpyruvate dioxygenase (HPPD). This enzyme plays a key role in the tyrosine degradation pathway. By blocking HPPD, nitisinone prevents the accumulation of toxic metabolites responsible for severe liver and kidney damage in some metabolic diseases. In simple terms, nitisinone acts as a biochemical traffic light, stopping the production of harmful by-products before they cause tissue damage.
Brand Names: Orfadin®, Nityr®, and Harliku® (new 2025 brand for AKU).
The Timeline of Nitisinone FDA Approval
2002 — First FDA Approval for HT-1
- The FDA approved Orfadin (nitisinone) on January 18, 2002.
- Indication: Treatment of hereditary tyrosinemia type 1 (HT-1).
- This marked a revolutionary moment, offering a non-surgical treatment for a previously fatal liver disorder.
2017–2020 — Formulation Advancements
- New formulations such as tablets and oral suspensions were introduced, improving ease of administration, especially in children.
2025 — FDA Approval for Alkaptonuria
- The FDA approved Harliku (nitisinone) for alkaptonuria (AKU) in June 2025.
- Indication: To reduce urinary homogentisic acid (HGA) in adults with AKU.
- This is the first FDA-approved therapy targeting the root biochemical cause of alkaptonuria.
This expanded indication has made nitisinone a cornerstone drug for multiple metabolic diseases.
Nitisinone FDA Approval Perspective
The FDA considered several critical aspects before approving nitisinone for AKU:
- Biochemical efficacy: Significant reduction in urinary HGA levels.
- Safety data: Acceptable safety profile with manageable tyrosine elevation.
- Clinical rationale: A clear connection between reduced HGA levels and slowed disease progression.
- Unmet medical need: No prior FDA-approved therapy for AKU existed.
This decision aligns with the FDA’s commitment to promoting orphan drugs for rare and underserved conditions.
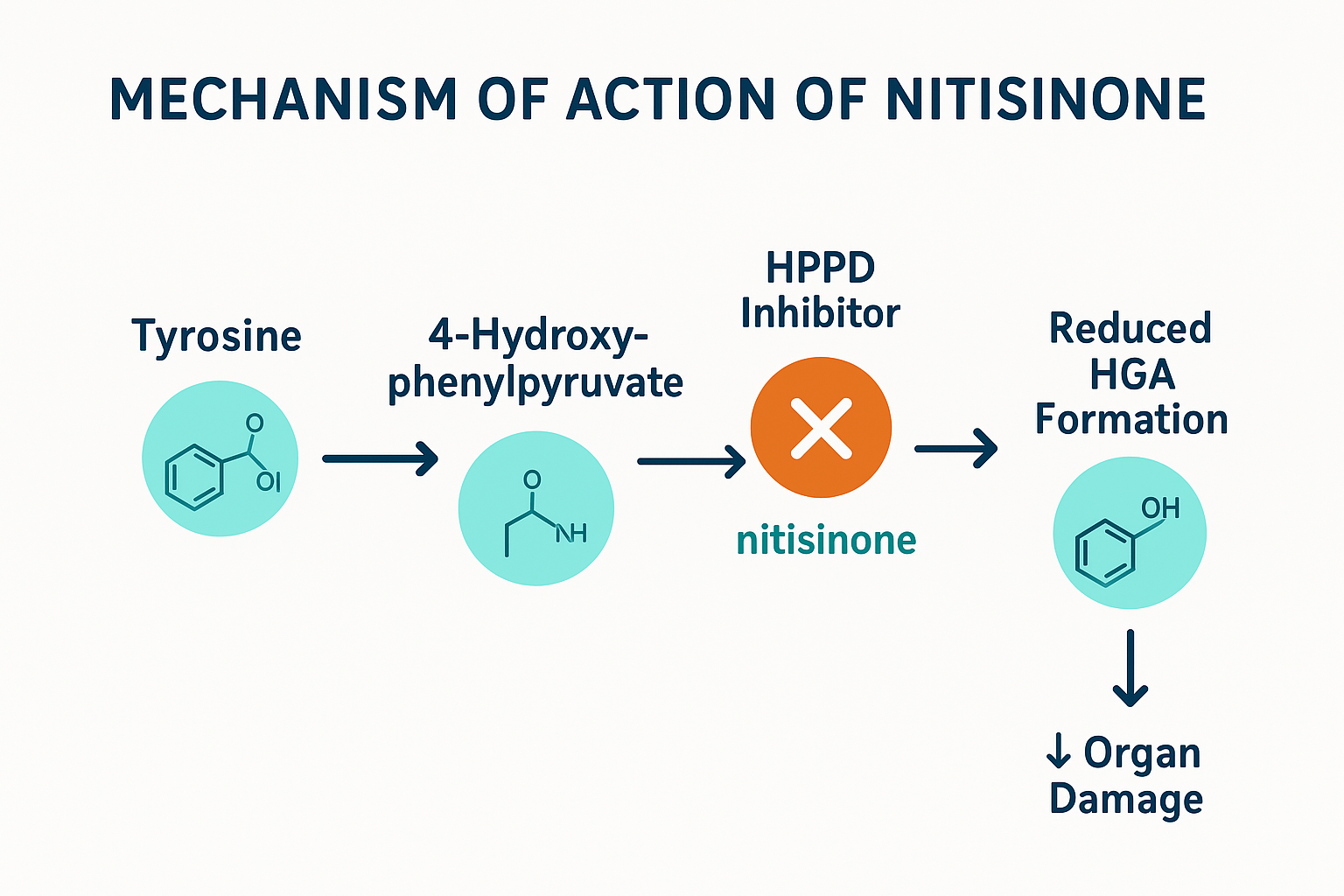
Mechanism of Action — How Nitisinone Works
Nitisinone inhibits the enzyme HPPD (4-hydroxyphenylpyruvate dioxygenase). This enzyme normally converts 4-hydroxyphenylpyruvate to homogentisic acid (HGA) – a step in tyrosine metabolism.
By inhibiting this step:
- The production of HGA is stopped.
- The accumulation of toxic metabolites that damage the liver, kidneys and connective tissue is prevented.
- However, tyrosine levels increase, which requires dietary control.
Approved Indications of Nitisinone
A. Hereditary Tyrosinemia Type 1 (HT-1)
- A rare inherited metabolic disorder affecting the breakdown of tyrosine.
- Untreated, it leads to severe liver failure, renal dysfunction, and neurological crises.
- Nitisinone + low-tyrosine/phenylalanine diet = significant improvement in survival.
B. Alkaptonuria (AKU)
- A chronic genetic disorder where HGA accumulates, leading to darkened urine, cartilage discoloration (ochronosis), and early-onset arthritis.
- FDA approved Harliku (nitisinone) in 2025 to reduce urinary HGA in adults.
Clinical Trials and Evidence Leading to Nitisinone FDA approval
For HT-1
Clinical data showed:
- Normalization of liver enzymes.
- Reduction in toxic metabolites like succinylacetone.
- Decreased need for liver transplantation.
- Improved long-term survival.
Long-term outcome studies confirmed that patients treated with nitisinone had dramatically higher 10-year survival rates compared to those untreated.
For AKU
In pivotal clinical trials:
- Nitisinone reduced urinary homogentisic acid (HGA) by over 95%.
- Improved joint and connective tissue health markers were observed in long-term follow-up.
- Safety profile consistent with earlier studies.
These results formed the basis for the FDA’s 2025 approval of Harliku.
Dosage and Administration
For HT-1
- Initial dose: 1 mg/kg/day, divided into two doses.
- Adjusted based on plasma succinylacetone levels.
- Must be combined with dietary restriction of tyrosine and phenylalanine.
For AKU (Harliku)
- Adult dosing as per label (typically fixed-dose tablet).
- Regular monitoring of:
- Plasma tyrosine levels
- Ophthalmologic status
- Urinary HGA
Note: Always follow the specific prescribing information and clinician guidance.
Safety and Side Effects
While nitisinone has a favorable benefit-risk profile, it requires careful monitoring due to elevated tyrosine.
Common Side Effects
- Eye symptoms (redness, photophobia, pain, corneal opacity)
- Skin irritation
- Nosebleeds
- Headache or fatigue
- Gastrointestinal upset
Serious Side Effects (rare)
- Elevated plasma tyrosine leading to corneal crystals or keratitis.
- Neurocognitive or mood changes (linked to high tyrosine).
- Reversible blood count abnormalities (rare).
Dietary management with low-tyrosine foods remains a key part of therapy to reduce risks.
Monitoring Requirements
Because nitisinone alters the metabolic pathway, ongoing monitoring is essential:
- Blood tests: Tyrosine and succinylacetone levels.
- Eye exams: Detect early corneal changes.
- Dietary review: Regular visits with a dietitian.
- Liver and kidney function tests: For overall metabolic health.
Benefits of Nitisinone Therapy
- Prevents life-threatening liver disease in HT-1.
- Reduces toxic metabolite accumulation in AKU.
- Improves long-term survival rates.
- Decreases need for liver transplantation.
- Enhances quality of life for patients with rare metabolic disorders.
The 2025 Nitisinone FDA Approval extends these benefits to a new population of adult AKU patients who previously had no effective therapy.
Economic and Access Considerations
As an orphan drug, nitisinone’s cost can be substantial. However:
- Manufacturer assistance programs (by Sobi and other partners) provide patient support.
- Insurance coverage has expanded following the new FDA indication.
- Generic versions in some regions may reduce long-term costs.
In the U.S., orphan drug status ensures market exclusivity and often drives future research investments.
Future Research and Expanding Horizons
Researchers are exploring:
- Long-term outcomes in AKU patients treated with nitisinone.
- Optimized dosing to reduce tyrosine-related side effects.
- Combination dietary and pharmacological regimens for improved outcomes.
- Early-diagnosis screening programs to start therapy sooner in HT-1.
Future label expansions or pediatric approvals may emerge as data accumulates.
The Role of Diet in Nitisinone Therapy
Since nitisinone raises tyrosine levels, patients must follow a strict low-protein diet that limits:
- Meat, dairy, nuts, and soy products.
- Processed foods high in tyrosine/phenylalanine.
A dietitian-supervised plan ensures safe nutrition while keeping tyrosine within a manageable range.
Global Impact and Regulatory Significance
The Nitisinone FDA approval 2025 signals a positive direction for rare-disease treatment:
- Encourages investment in metabolic research.
- Sets a precedent for expanding older orphan drugs to new indications.
- Demonstrates how real-world data supports label expansion decades after initial approval.
Other agencies such as the EMA (Europe) and Health Canada are reviewing similar submissions, paving the way for global harmonization of nitisinone’s indications.
Why the 2025 Nitisinone FDA Approval Matters for Patients
Before Nitisinone FDA approval , AKU patients had no FDA-approved drug targeting the underlying cause of their condition. Treatments were purely supportive — addressing pain, inflammation, and joint replacement in advanced stages.
Now, with nitisinone FDA approval for AKU, patients can:
- Begin disease-modifying therapy early,
- Prevent or delay cartilage and joint degeneration,
- Experience improved quality of life through biochemical control.
This is a major milestone not just for metabolic medicine, but for all rare disease communities.
Key Takeaways
- First approved in 2002 for HT-1.
- Expanded approval in 2025 for AKU (Harliku).
- Mechanism: HPPD inhibition → reduced toxic metabolites.
- Requires dietary control and regular monitoring.
- Represents a landmark achievement in rare disease therapy.
Final Thoughts — The Legacy of Nitisinone
Nitisinone’s journey to Nitisinone FDA Approval exemplifies how scientific perseverance, patient advocacy, and regulatory cooperation can revolutionize the treatment of rare diseases.
From saving the lives of children with HT-1 to giving hope to adults with alkaptonuria, nitisinone stands as a symbol of medical progress and precision-based metabolic therapy.
As post-approval studies continue, the global medical community awaits deeper insights into how this changes the long-term outlook for these once-neglected disorders.

