Table of Contents
Introduction
Prostate cancer is one of the most common malignancies affecting men worldwide. Despite major advances in hormonal therapy, resistance to conventional treatments such as enzalutamide and apalutamide limits long-term efficacy. Enter Rezvilutamide – a next-generation androgen receptor inhibitor that is redefining how doctors approach prostate cancer therapy.
Developed as an innovative treatment option for metastatic castration-resistant prostate cancer (mCRPC), Rezvilutamide is garnering global attention for its improved efficacy, better tolerability, and reduced central nervous system (CNS) penetration compared to previous drugs in its class.
This comprehensive guide explores everything you need to know about Rezvilutamide – its mechanism of action, pharmacokinetics, clinical efficacy, safety profile, and future potential in prostate cancer management.
What Is Rezvilutamide?
Rezvilutamide is a novel androgen receptor (AR) antagonist developed by Suzhou Kinter Pharmaceuticals for the treatment of prostate cancer, particularly mCRPC and metastatic hormone-sensitive prostate cancer (MHSPC).
Unlike traditional anti-androgens, Rezvilutamide exhibits high selectivity and affinity for androgen receptors, effectively inhibiting tumor growth driven by androgen signaling. It belongs to the same pharmacological class as enzalutamide but has structural modifications that increase its therapeutic index and reduce CNS-related side effects.
The chemical design of Rezvilutamide aims to overcome the resistance mechanisms commonly seen in long-term androgen deprivation therapy, providing a more potent, safer, and better tolerated therapeutic option for patients.
Mechanism of Action
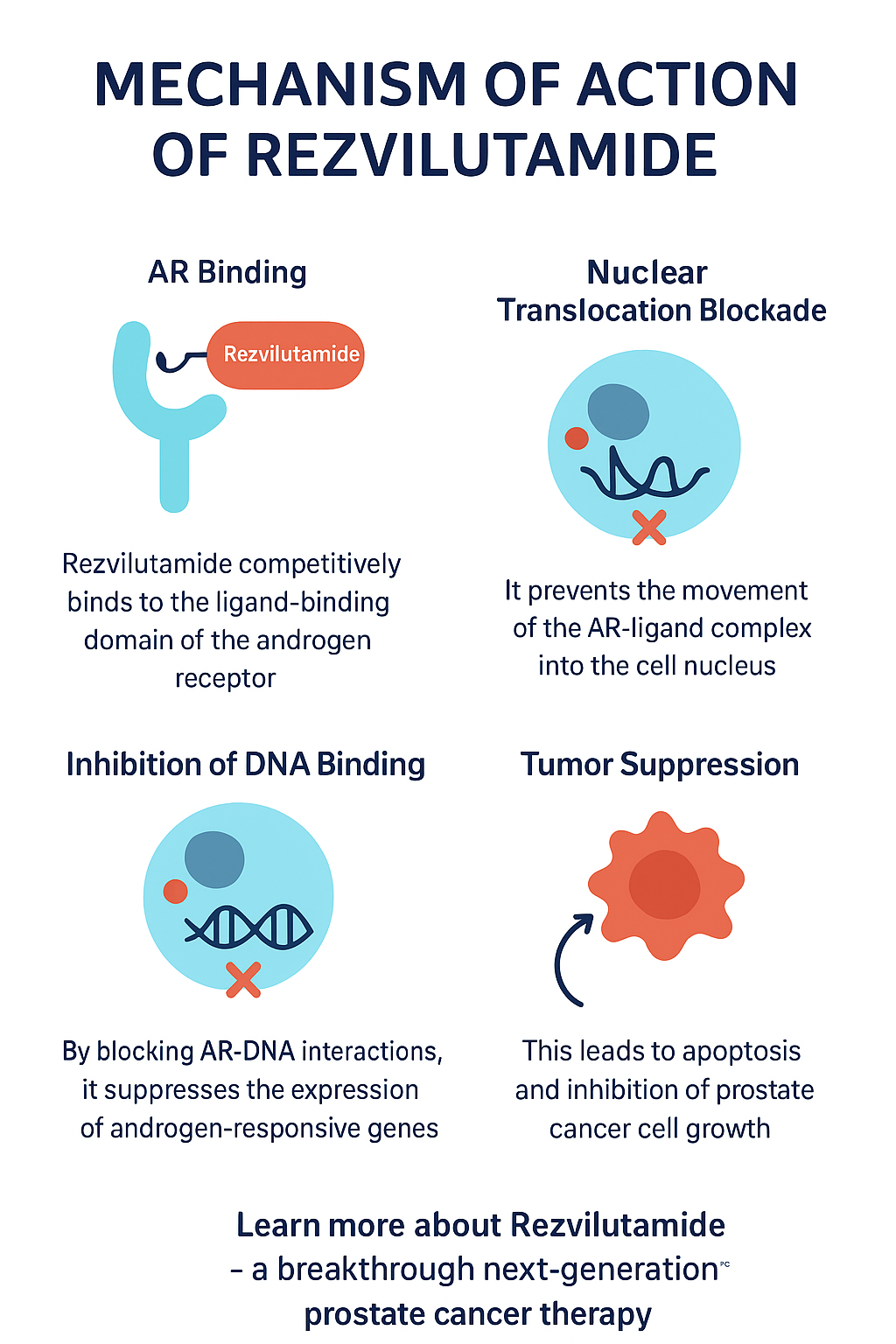
The mechanism of action of Rezvilutamide is centered on its potent inhibition of androgen receptor signaling, which plays a key role in the progression of prostate cancer.
1. AR binding: Rezvilutamide competitively binds to the ligand-binding domain of the androgen receptor, preventing natural androgens such as testosterone and dihydrotestosterone (DHT) from activating it.
2. Nuclear translocation blockade: It prevents the translocation of the AR-ligand complex into the cell nucleus, a key step required for DNA binding and gene transcription.
3. Inhibition of DNA binding: By blocking AR-DNA interactions, Rezvilutamide suppresses androgen-responsive gene expression involved in cell proliferation and survival.
4. Tumor suppression: This cascade causes apoptosis (programmed cell death) and inhibition of cancer cell growth, slowing or stopping the progression of the disease.
Simply put, resveratrol reduces the “fuel” – androgenic stimulation – that prostate cancer cells need to grow, but it doesn’t affect the central nervous system as much as older agents.
Pharmacokinetic Profile
Rezvilutamide’s pharmacokinetic (PK) characteristics play an important role in its improved efficacy and safety:
- Oral Bioavailability: High, enabling convenient oral dosing.
- Half-Life: Moderate, allowing once-daily administration.
- CNS Penetration: Significantly lower compared to enzalutamide, reducing the risk of CNS-related side effects such as seizures or cognitive impairment.
- Metabolism: Primarily hepatic, involving cytochrome P450 enzymes.
- Excretion: Mainly via feces, with minimal renal elimination.
This pharmacological optimization contributes to steady plasma concentrations, consistent anti-tumor activity, and fewer adverse effects.
Clinical Uses
It is primarily being developed and evaluated for the following clinical indications:
1. Metastatic Castration-Resistant Prostate Cancer (mCRPC)
In patients whose cancer continues to progress despite castration-level testosterone, rezvilutamide has shown promising results in delaying disease progression and improving overall survival.
2. Metastatic Hormone-Sensitive Prostate Cancer (mHSPC)
Ongoing clinical trials suggest that rezvilutamide could be beneficial in earlier stages of prostate cancer, especially in combination with androgen deprivation therapy (ADT).
3. Non-Metastatic CRPC (nmCRPC)
Preclinical and early-phase trials indicate potential in non-metastatic CRPC cases, aiming to prevent the transition to metastatic disease.
Rezvilutamide vs. Enzalutamide: Key Differences
| Feature | Rezvilutamide | Enzalutamide |
| Generation | Next-generation AR inhibitor | First-generation AR inhibitor |
| CNS Penetration | Low | High |
| Risk of Seizures | Minimal | Moderate |
| Selectivity | Higher AR selectivity | Moderate |
| Efficacy | Improved progression-free survival | Proven but limited by resistance |
| Tolerability | Better | Slightly poorer |
| Pharmacokinetics | Stable and predictable | Variable with long half-life |
It’s reduced CNS activity is a major advantage, particularly for elderly patients or those with comorbid neurological conditions.
Clinical Trial Insights
Multiple phase II and III trials have evaluated the safety and efficacy of this drug in prostate cancer patients.
1. Phase III Trial (ARADES-001)
- Population: Men with mCRPC previously treated with ADT.
- Findings: Rezvilutamide significantly prolonged radiographic progression-free survival (rPFS) compared to placebo.
- Outcome: Statistically significant improvements in PSA response and tumor control rate.
2. Phase IIb Study
- Design: Compared rezvilutamide with enzalutamide in terms of CNS side effects and efficacy.
- Result: Rezvilutamide showed similar tumor control with markedly fewer neurological adverse events.
3. Ongoing Global Trials
Further investigations are assessing rezvilutamide in combination with ADT, abiraterone, and chemotherapy to enhance outcomes and overcome resistance in advanced prostate cancer.
Benefits
- Enhanced Selectivity: Targets androgen receptors more precisely.
- Improved Safety Profile: Lower CNS side effects such as fatigue, dizziness, or seizures.
- Better Tolerability: Suitable for long-term therapy.
- Potent Anti-Tumor Activity: Effective against resistant prostate cancer cells.
- Convenient Oral Dosing: Once-daily administration enhances adherence.
- Combination Potential: Can be safely combined with standard hormonal or chemotherapeutic regimens.
These benefits position rezvilutamide as one of the most promising new-generation anti-androgens for managing prostate cancer.
Side Effects
Although generally well-tolerated, rezvilutamide may cause certain adverse effects. Based on clinical studies, common and rare side effects include:
Common Side Effects
- Fatigue
- Decreased appetite
- Nausea
- Hot flashes
- Arthralgia (joint pain)
- Mild liver enzyme elevation
Less Common or Serious Side Effects
- Hypertension
- Anemia
- Rash
- Elevated bilirubin levels
- Rare CNS effects (e.g., mild headache or dizziness)
Overall, rezvilutamide side effects are notably less severe than those of enzalutamide, especially concerning CNS toxicity.
Dosage and Administration
- Formulation: Oral tablet
- Typical Dose: 160 mg once daily (subject to clinical protocols)
- Administration: Taken at the same time each day, with or without food
- Monitoring: Regular liver function tests, PSA levels, and blood counts recommended
The simplified dosing regimen supports better patient compliance and consistent therapeutic response.
Recent Research and Developments
In recent years, this drug has gained recognition not only for its therapeutic potential but also for its role in combination therapy and early-line treatment.
Key highlights include:
- 2023–2025 studies demonstrating superior PSA reduction rates.
- Promising results in Asian and global populations, showing consistent efficacy.
- Ongoing collaborations exploring rezvilutamide + abiraterone acetate combinations to delay resistance.
Researchers are also investigating rezvilutamide’s potential beyond prostate cancer, including certain androgen receptor–driven tumors, though such applications remain experimental.
Future Prospects
The future looks bright for rezvilutamide in the landscape of oncology. With positive outcomes in pivotal trials and expanding global awareness, this drug could soon become a standard of care for advanced prostate cancer.
Potential future directions include:
- FDA and EMA approvals for global use.
- Combination regimens with immunotherapy or PARP inhibitors.
- Use in early-stage prostate cancer or high-risk localized disease.
- Development of biomarker-driven treatment algorithms to personalize rezvilutamide therapy.
Given its unique pharmacological profile and consistent clinical success, it may soon redefine the treatment paradigm for androgen receptor–dependent cancers.
Conclusion
Rezvilutamide is at the forefront of prostate cancer treatment – a next-generation androgen receptor antagonist that offers strong efficacy with improved safety. Its low CNS penetration, high selectivity, and consistent clinical results make it a strong candidate to replace or complement existing anti-androgen treatments.
As research continues, it may soon emerge as a cornerstone treatment, offering new hope to patients battling advanced prostate cancer around the world.
FAQs
1. What is rezvilutamide used for?
2. How does rezvilutamide differ from enzalutamide?
3. What are the most common side effects of this drug ?
Disclaimer
The information provided in this article about Rezvilutamide is intended for educational and informational purposes only. It should not be used as a substitute for professional medical advice, diagnosis, or treatment. Always consult your healthcare provider or a qualified medical professional before starting or changing any medication or treatment plan. While every effort has been made to ensure the accuracy of this information, new research and clinical data may update current knowledge about Rezvilutamide. The author and website assume no responsibility for any potential consequences from the use of information contained herein.